Autism spectrum disorders and DNA testing
Dr.M.Raszek
Genetics dominate the world of autism spectrum disorders
Merogenomics recently attended the annual human genomics conference put on by the American Society of Human Genetics. We liked the online style because it allowed us to really take the time to explore all the different topics. One area of interest with multiple talks was dedicated to the genetics of autism and so we took the opportunity to delve into this important topic.
Everyone knows how the incidence of autism has been on a constant rise over the years (currently approximately one in 54 children in the US and one in 66 in Canada). But what is not broadly understood in the general public is that genetics play a massive contribution in autism spectrum disorders. In turn, it is also not widely known that identifying the genetic factors in an affected autistic individual can help to more specifically discriminate the condition. This is because an autism diagnosis is made based strictly on behavioural features without the need to understand what is actually causing these symptoms. Consequently, genetics does not have to play a role in diagnosis.
But DNA testing certainly can play a very important role in helping with the proper management of autism, and the main reason why is because targeted genetic identification of autism spectrum disorders could help clearly define risks of additional health problems that could be associated with that specific autism. This can be extremely valuable because it allows for proactive screening to look for the development of these additional health problems (they are referred to as comorbidities, a term you might have now come across with regards to COVID-19). And when discovered early, they can lead to better management of the cases. This is by far the largest benefit of any genetic screening of autistic individuals.
DNA testing to the rescue for autism?
On top of that, autism is known to be very heterogenous in nature, meaning it has very broad array of symptoms, and so it is indeed often identified with other comorbidities. It is very well recognized now that autism often links genetically with other neurodevelopmental conditions. Once again, if genetically determined then the precise understanding of what these additional conditions might be could be used to positively affect patient management.
There is also a slight possibility that certain autism cases might actually be misdiagnosed cases of metabolic disorders, and so if these cases were identified with a genetic screening, it could lead to corrective treatments. However, this is a rare occurrence with likely less than 1% of autistic kids actually suffering from metabolic disorders instead.
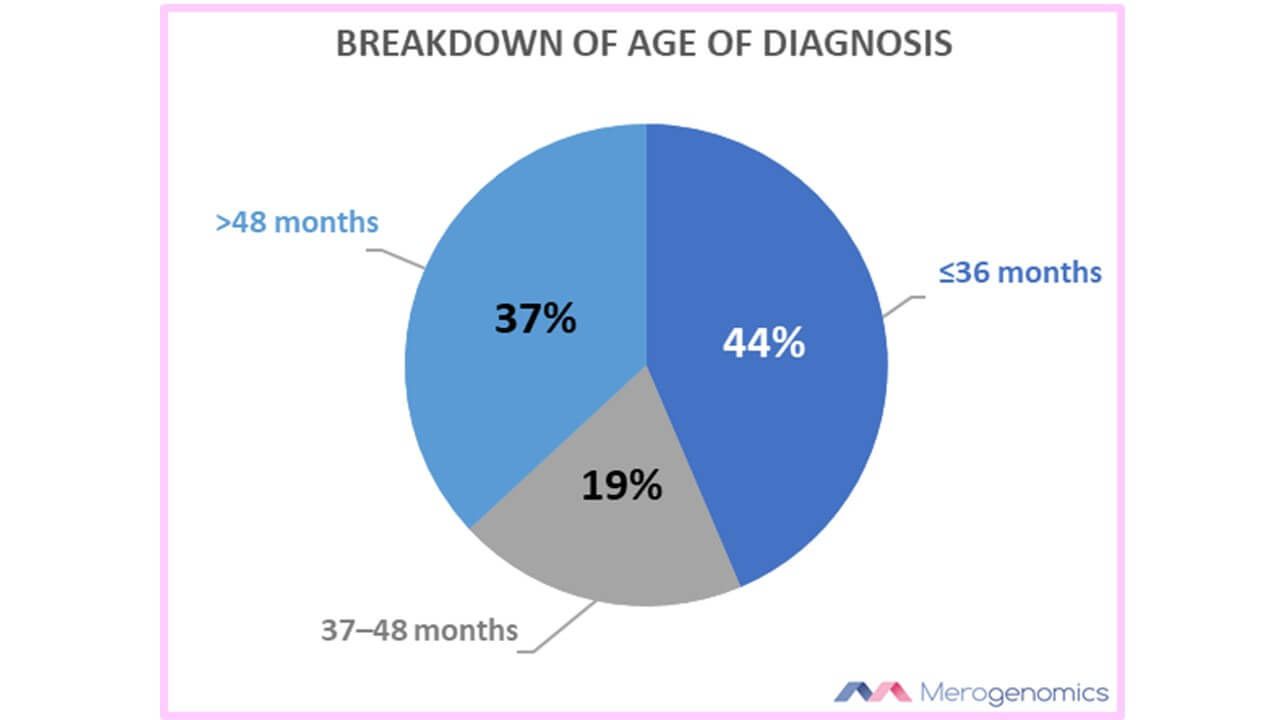
Genetic testing could also in theory speed up the process of a diagnosis if known genes linked to autism spectrum disorders were identified in a child. In the USA the median age of first evaluation of autistic children is 40 months of age, with the diagnosis at 51 months of age. It is significantly earlier for autistic children with comorbid cognitive impairment (also described by others as intellectual disability). The point being that the condition might not be easily recognized early on if not accompanied by certain, obvious recognizable features. Suspicions can be rapidly confirmed in instances where genetic involvement is uncovered by testing. This is also important because there is mounting evidence that early behavioural intervention in autistic children can improve a child’s cognitive, language and adaptive skills.
Finally, DNA testing can also be used towards a better understanding of the use of medications for the treatment of co-occurring psychiatric and behavioural comorbidities, which is truly the main area of therapy available for individuals diagnosed with autism (there are no curative treatments for the core symptoms of autism). This is referred to as pharmacogenetic DNA testing (often also described as pharmacogenomic testing although there is distinction between the two terms), a topic we have previously covered extensively. For an overview of pharmacogenetic DNA testing you can watch the Merogenomics video below.
This is quite important because a majority of children with autism spectrum disorder will be prescribed some medication by the time they reach their adolescence. For example, the only two FDA approved medications for use in autism spectrum disorders have a pharmacogenetic link, meaning that their use can be effectively driven through an understanding of the patient’s genetic information. This is potentially quite significant as these drugs can and do produce unwanted side-effects. One of these is weight gain, which in turn is managed with the metformin drug, which itself also has pharmacogenetic associations.
Pharmacogenetic association is also linked to certain treatments of attention deficit hyperactivity disorder, obsessive-compulsive disorder symptoms, as well as anxiety, depression and/or agitation - all of which are potential symptoms treated in autism. In fact, of the 26 medications rated by families affected with autism spectrum disorder for effectiveness in symptoms treatment, currently 21 can be linked to personal genetic information. However, it needs to be noted that at the moment there is no clinical trial data confirming the benefits of pharmacogenetic screening in specifically autistic individuals. But at the very least, pharmacogenetic testing might be of value when a person has attempted to try multiple medications to treat any given symptom.
Autism genetics brief overview
Before we move to the discussion of presentations from the 2020 ASHG conference, it is also important to stress how often genetic causes are identified in those diagnosed with autism. One estimate suggests that genetic causes of autism can be identified in approximately 35% cases. Autism is a very complex interplay between genetics and environment, and likely this figure is bound to change as more research comes out and is clinically validated. Currently there are literally hundreds of different genes that have been linked to autism (it is estimated that over a 1000 could have contributing factors), but only a subset of these are actually validated for meaningful medical investigation. In addition, the types of genetic alterations that can contribute to the condition’s development can also be incredibly varied, adding to the complexity of the resulting physical traits. In addition, the diagnostic yield, or the rate of success of genetic testing, increases for cases with cognitive impairment or other syndromic features (features indicative of additional diseases). In such cases, genetic testing should always be considered which might not necessarily be the current practice. While the recognition of DNA testing in autism is increasing, we have also visited doctors that specialize in the management of autistic spectrum disorders where genetics does not come into play at all for them.
In addition, there are multiple genetic testing options available to accommodate the identification of different genetic alterations that are encountered. These can include single point mutations (DNA mutations affecting only one nucleotide within the DNA code) to complex structural variants. One common type of alteration is what is referred to as copy number variations, where a segment of DNA material is either duplicated multiple-fold times, or some of it is lost instead. You can see how that could be damaging, as such an event could either dramatically increase or reduce the amount of a specific protein being produced which then could throw off the balance of biological functions within cells and organs. To complicate matters further, many of the mutations (properly referred to as variants) are not inherited from parents at all, but arise spontaneously in the gametes that were used to create a new baby. These are referred to as de novo variants (or mutation of new origin). As a consequence there might not be a one-size-fits-all type of DNA test that could provide all the answers (with perhaps full genome sequencing being the closest alternative), and typically clinical analysis of DNA from autistic patients requires use of multiple technologies to get the best chance of discovery.
The final type of DNA variants that can contribute to autism is what are referred to as somatic variants, or basically mutations that arise in any cell that is not a gamete, so in any cells that are not sperm or eggs. Somatic variants can occur in any cell, anywhere in the body throughout the entire lifespan, although of course these somatic mutation events that contribute to autism development have to occur in the earlier stages of brain development to impart its impact. As a consequence, somatic mutations are much harder to track down because these mutations are only present in a subset of cells, and not in every single cell of the body (unlike those inherited or de novo variants). Watch a brief Merogenomics video on somatic mutations below.
Mega studies to understand autism
What is amazing is that to be able to identify the sheer number of genetic markers that could be influencing autism development, studies that include literally thousands of patients with autism (whose DNA is being sequenced) are currently taking place! A major such endeavour is SPARK, which aims to collect data on 50,000 families with more than 25,000 families already enrolled and more than 10,000 autistic children already DNA tested. Such extremely large studies allow the discovery of even very rare contributing factors to the disease with a significant statistical power. It is such large studies, costing millions of dollars, that help to shape the medical understanding of genetics, and continuously grow the power of DNA sequencing interpretation as it relates to human health. Once you sequence your genome, you could in theory be re-interpreting it for the rest of your life (which Merogenomics advocates).
Here is how SPARK breaks down the different types of genetic factors contributing to autism.
As you can see, genetic contribution is considered to be the bulk of what drives the development of autism spectrum disorders, although this figure was generated from variety of different reference materials. But one category that is interesting is referred to as the loss-of-function mutations. You can probably figure out what that means. It means that the mutation prevents a specific gene from being used to produce a functional protein. Recall from our last blog post that DNA is only a map of information which then has to be copied into a blueprint (RNA) which is then used to produce proteins, or the molecular robots that do all the dirty hard work around the house (cell). That is, by the way, the definition of a gene - a segment of DNA that is used for the eventual production of a protein. Thus loss-of-function mutation means a mutation that prevents a functional protein from being built. It is just a subcategory of mutation, so it can be inherited, it can be de novo or of somatic in origin. Sometimes they are without an effect (the body makes up for the resulting impact on protein production) and sometimes they can be deleterious, and lead to a disease (reduced or lost quantity of protein is detrimental).
Loss-of-function mutations are especially common in autism and they are observed more frequently than in a broader population. And most often they are of the de novo nature, meaning neither of the parents actually had this mutation and it arose spontaneously in the sperm or an egg genetic material. Now let’s give you a taste of the type of information that is being uncovered and was presented at the ASHG 2020 conference. One of the talks presented by Dr. Xueya Zhou based on the SPARK study results discussed an interesting case of loss-of-function mutation in the UBR1 gene that increased risk of autism development but only if it was inherited! For some reason the gene was never affected by de novo events. Meaning, the mutation could not just show up spontaneously in the sperm or the egg, it had to be present in the genome of one of the parents. It is the first such identified gene that increases the risk of autism only as inherited loss-of-function mutation, and therefore it is an interesting oddity. It shows you just how complex the world of genetics can be. Another fascinating talk also focusing on de novo mutations in autism, specifically discussed looking for such mutations in the regions of DNA that are not used as the map to produce proteins. Recall that segments of DNA that are used to produce proteins are called genes. Genes actually comprise only about 2% of all of your DNA (or 2% of your genome). All of the rest of the DNA is referred to as non-coding (as in it does not code for protein production) and is used for DNA management purposes with a bit of historic evolution garbage left behind here and there with potentially no good purpose (formerly referred to as junk DNA, but we are learning that the biggest junk here is merely our ignorance).
In this case, autism de novo mutations in a non-coding region of DNA was found, in a region referred to as a promoter. Promoters are regions of DNA recognized by proteins that are used to copy DNA into small RNA fragments. Such protein robots are called transcription factors (transcription is the name for the process of copying DNA into RNA). What was elegant about this story is that this promoter, called hs737 (scientists and airline industry folks must be hanging in the same bars) happens to also be frequently affected in other neurodevelopmental disorders besides autism, through copy number variation. Recall that copy number variation is just some science slang for a segment of the DNA being replicated in some quantity. If such a copy number variation is affecting a gene, it can influence the final amount of the gene product, i.e. the amount of some protein. And that is exactly what was observed with hs737, affecting a quantity of a specific protein in these neurodevelopmental disorders. Furthermore, de novo mutations in the gene coding for that same protein are also observed in neurodevelopmental disorders, including hypotonia, ataxia and delayed development syndrome (HADDS). And guess what? The autistic cases who exhibited the DNA mutation in the hs737 promoter also showed hypotonia (low muscle tone). This research story is an excellent example of how understanding the genetic background can provide an understanding of resulting autism traits.
What can you expect with DNA testing in autism?
But our favourite talk dedicated to autism at the conference was by Dr. Jessica Wright - who also presented on behalf of the SPARK consortium - with what is likely the best real-life scenario of what families with autism could experience if they undergo genetic testing. The information presented was based on the testing of nearly 30,000 individuals including about 10,000 individuals with autism who were screened for a variety of mutations in over 150 genes in addition to copy number variations in nearly 30 additional genomic regions. The complete list of investigated autism related genes is available on the SPARK website. The selected genes are highly verified and expected to be more on the harmful and highly penetrant side (meaning genes highly likely to be contributing to a disease manifestation, see our previous post for a good explanation of disease penetrance). This list is expected to continue to grow as more data keeps emerging but as a consequence of this specific selection of genes, it is expected that the frequency of the detection of mutations might be reduced. But if a family of an autistic child were undergoing screening right now, with a bona-fide clinical DNA test, this is likely what they would experience.
We contacted SPARK to see if we could present this latest data for you and they kindly gave us permission. The rate of return of results overall was 8% for all the tested autism spectrum disorder individuals, with just over half of these being of de novo nature. However, the more complex the presentation of autism was, the more likely that some genetic findings would be returned to a family. Thus 12% of autistic individuals with cognitive impairment, 18% of autistic individuals that experienced seizures, 21% if the autism was also associated with being non-verbal and 22% of autistic individuals afflicted with birth defects obtained genetic results. On the other hand, autism with above average cognitive abilities had the lowest rate of return of results, with 4%.
These are similar results to what has already been published based on the SPARK research of a much smaller cohort - just over 450 families. In that publication, the return of genetic results was 10% for all of the tested autism spectrum disorder individuals. We wanted to bring this up because in that study if the family had more than one affected child, the discovery rate of genetic finding also increased, to 15%, which we also found very informative.
To conclude, the take home message is that genetic analysis of individuals with autistic spectrum disorders can yield results with potential benefits to families and should always be considered especially in cases with additional complications. Genetics play a very large role in autism development, and over a hundred genes are already linked to it, and it is expected that this will continue to grow, especially with the discovery of DNA mutations of small impact (mutations that are not highly penetrant). However, these small impact mutations can be used towards the development of polygenic risk scores which will also be expected to help understand the risks of associated comorbidities. The other benefit of DNA testing can be linked mainly to the use of medication which is common in the treatment of autism comorbidities. We expect these benefits to grow over time so in the meantime, we hope more doctors will turn to DNA testing in autism.
This article has been produced by Merogenomics Inc. and edited by Jason Chouinard, B.Sc. Reproduction and reuse of any portion of this content requires Merogenomics Inc. permission and source acknowledgment. It is your responsibility to obtain additional permissions from the third party owners that might be cited by Merogenomics Inc. Merogenomics Inc. disclaims any responsibility for any use you make of content owned by third parties without their permission.
Products and Services Promoted by Merogenomics Inc.
Select target group for DNA testing
Healthy screening |
Undiagnosed diseases |
Cancer |
Prenatal |
Or select popular DNA test
 |
 |
 |
 |
Pharmaco-genetic gene panel |
Non-invasive prenatal screening |
Cancer predisposition gene panel |
Full genome |