Pregnancy screening options and the role of NIPT
Dr.M.Raszek
One oocyte but two sperms?
Recently a string of unusual pregnancy cases have been reported, which are so atypical in some instances that they were thought to be impossible!
Twins are traditionally classified as monozygotic or dizygotic. Monozygotic twins are identical twins with identical genomes and are a result of a single fertilized egg (oocyte) that divides into two during the early development of the future embryo. Dizygotic twins, or fraternal twins, result from two separate eggs each fertilized by independent unique sperm cells. Dizygotic twins share approximately 50% of their DNA sequence identity, just as any siblings do. Because identical twins form due to the division of cells early after the fertilization takes place, monozygotic twins can share a placenta. In fact, the majority of identical twins (75%) share a placenta.
The placenta is the attachment site by which the embryo in its protective sac is fused to the mother’s uterus, and is the site of exchange for all of the molecular information between the mother and the baby. The baby’s blood circulates into the placenta, down to tree-like structures called chorionic villi, which in turn are washed by mother’s blood. The mother’s blood provides everything a baby needs, and removes everything that the baby needs to dispose of, without actually mixing blood supplies. But the layer of cells that separate the mother’s blood on the outside of chorionic villi from the baby’s blood on the inside is thin enough that all small molecules can pass from mom to the baby. So with every single beat of the mother’s blood, she nourishes and cleans the baby, and protects it from harm.
The placenta develops from the same fertilized egg that produces the embryo. This means it should be genetically identical to the baby. It also means that fraternal or dizygotic twins, which come from two independently fertilized eggs, will produce their own placenta to connect and interact with the mother. This in turn means that if you see twins sharing a placenta on an ultrasound, it automatically indicates that they are identical twins.
However, recently this dogma has been put to the test with a woman who showed on the first ultrasound twins sharing one placenta, but then on a subsequent ultrasound it was revealed that the twins were two separate genders! That’s next to impossible if they are supposed to be identical twins, although there could always be a genetic mutation on a Y chromosome preventing the chromosome from performing its proper function towards biological gender development. It turned out that these twins were an ultra-rare case of sesquizigotic twins.
What are sesquizigotic twins? They are twins produced from one egg that was fertilized by two separate sperm cells – I bet you didn’t know that was even possible! Don’t worry, until recently, no one did. Basically what happens is that after fertilization with two sperms, the resulting cell had three sets of genomes, one from the mother and two from the father, that were different from one another (each sperm cell or egg cell is a random mixture of DNA, so each one of these oocytes or sperm cells comes with a unique genome). This normally would not be viable nor able to produce a baby, but the system fixed itself by first duplicating all of that DNA – so now there were six genome copies, two maternal and four paternal – and then dividing it correctly to daughter cells (two cells with right amount of maternal and paternal DNA, and one cell with right amount of only paternal DNA). Two of these new daughter cells resulted in future babies. The one with only paternal DNA could not continue with a normal existence, so it threw in the towel on its uniparental existence.
In the end, the two twins were identical for the maternal component of their genomes, but 78% identical for their paternal DNA!
The first such example of twins with the same maternal genome but mixed paternal contribution, with about 50% of shared father genome in each twin, had been reported just in 2007. In that example, the birth of a true hermaphrodite (an ambiguous gender with the development of both ovarian and testicular elements), and a boy was described. But in that case, no placenta was examined, and the ultrasound records could not identify if the twins actually shared same placenta.
Prior to that, a similar event was described in 1998 to explain the genetics of a single hermaphrodite baby/. So these were very rare events indeed, at least in terms of being noted and reported.
Another recently described rare event was the birth of a baby with its partially developed twin growing right inside! Reported by The New York Times, it is uncertain if this case will see formal scientific publication, but a concrete genetic investigation on both the surviving baby and the underdeveloped twin is to take place. The occurrence is believed to happen in approximately 1 in 500,000 pregnancies, recounted only in about 200 cases thus far worldwide. There is not even a fully confirmed understanding on how this occurs, but it is believed that one fetus could be fully enveloping the other. For added dramatic effect, these cases usually are described as a baby being pregnant with its own twin, which is obviously quite inaccurate, but definitely eye catching!
These are ultra-rare events, so sparse that they make medical history when uncovered and reported. This is not what a pregnant woman has to even remotely be concerned about. There are, however, much more common events related to pregnancy that could require more serious attention.
Pregnancy outcomes
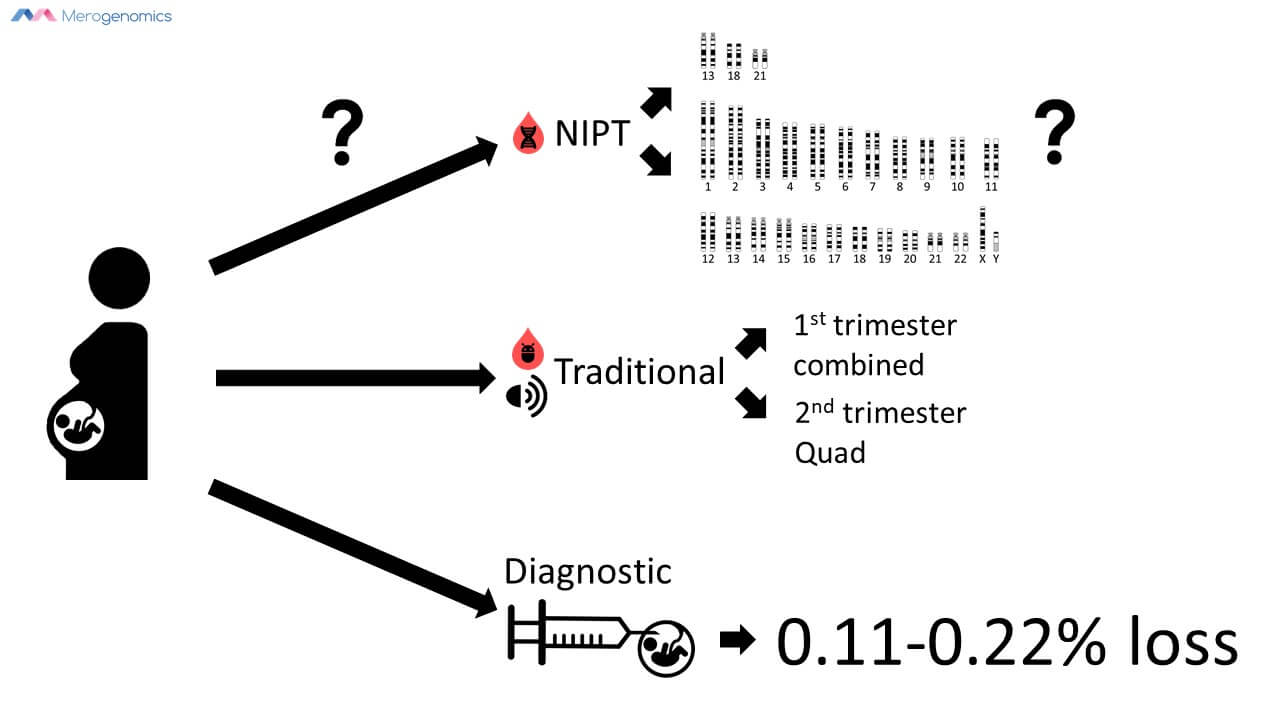
For the sake of simplicity, we will divide the pregnancy experience into four potential outcomes: normal and uncomplicated pregnancy, which can be expected in about 95% of pregnancies, and then other outcomes that we’ll divide into three main categories here. Number one is chromosomal abnormalities, which will remain the focus of this article as they account for the largest likelihood of experiencing pregnancy-related problems. What is referred to here are any problems related to having additional or deleted chromosomes in the developing fetus, whether in whole or in part. For the background on genome and chromosomes you can check out this past post. The most common problem encountered that we currently know of is that of an extra chromosome 21, resulting in the presence of three chromosomes 21, or trisomy 21, colloquially referred to as Down syndrome. This group specifically comprises the problems that already pregnant women can easily monitor for with non-invasive prenatal testing (NIPT) which we covered previously in detail. There is a limit to what can be detected with NIPT, and typically the smallest fragments of chromosome monitored with NIPT would involve at least a million nucleotides within the DNA code (nucleotides are the molecules that are the building blocks of the DNA).
The second group of potential problems encountered with the developing baby is are genetic diseases. In this case, the problems occur from mutations in the DNA that affect either a single specific site (single nucleotide), to millions of nucleotides. Typically this group would not be detected with NIPT, with the exception of some of the small chromosomal alterations mentioned above (but which are still rarely tested for). Therefore, if the pregnancy is suspected to be impacted by a specific genetic condition, the only way at the moment to test for this would require an invasive method of collecting the DNA sample of the fetus, either a chorionic villi sample, or amniotic fluid directly from the sac encasing the baby. This procedure is referred to as “amniocentesis” and is the only way to directly collect actual fetal cells (as opposed to chorionic villi sampling which will grab placenta cells). Once you get hold of those cells, the sky is the limit and you could even do a full genome scan, so you can diagnose practically any genetic disease provided that enough sample DNA is available. This service is completely available if needed.
The final possibility we will list here are the isolated birth defects. These are complex events that are a combination of both a multitude of genetic factors and environmental influence, so there could be hundreds of different causes behind their formation. Because of these multifaceted factors involved, there is no way to screen for this with any specific method. There are simply too many unknown variables involved to know what to screen for. At the moment, the best way to attempt to capture any of these events during pregnancy would be through a second-trimester ultrasound that is part of the typical obstetrics care already. At that point, if there are any congenital deformities, hopefully skilled staff will be able to detect it. Unfortunately, many of the developmental defects will still not be present or found at this stage of the fetal development. But this is one of the reasons why that later ultrasound screening is so important.
The physiological outcomes of all these events can be very diverse, ranging all the way from an apparently clinically normal baby, to a wide spectrum of intellectual disability, developmental delay, differences in body structure, and general impairments to many body systems and functions, all of which can negatively impact lifespan duration and even post-birth survival. According to an amazing March of Dimes 2006 Global report on birth defects, in Canada, in total, these three groups account for 4.6% of all pregnancies that result in the live birth of a child with birth defects.
The March of Dimes report defined birth defects as “abnormalities of structure or function, including metabolism”, and therefore might not include obvious physical changes at birth, and include around 7000 such conditions in this group. This sounds like basically all known genetic disorders. However, there are estimates that nearly 10% of the population could have health-related genetic conditions, and there is some preliminary data to support this. So the difference in these numbers might be what can be detected at birth or soon after birth versus what conditions can remain undetected, sometimes for years (such as cancer predispositions for example).
Nevertheless, the March of Dimes report lists the five most common birth defect conditions to be congenital heart defects, neural tube defects, hemoglobin disorders (thalassemia and sickle cell disease), Down syndrome (trisomy 21) and glucose-6-phosphate dehydrogenase (G6PD) deficiency, which together account for 25% of all of birth defects. Congenital heart defect is a good example of a birth defect that can be caused by many different genetic mutations, and some of these could be diagnosed with prenatal DNA sequencing.
Pregnancy screening options: a mother’s choice
The primary concern and screening available for women is that for chromosomal abnormalities. Until the recent introduction of NIPT, the screening program consisted of testing for specific blood markers (whether protein molecules or smaller chemicals), and an ultrasound, in the first and second trimesters of pregnancy. While there is a wide array of possible tests available, the most common traditional screening options are first-trimester combined screening, which looks for a couple of blood markers and includes an ultrasound (for nuchal translucency measurement), between 11-14 weeks of gestation, or a Quad test (so-called because it looks for four different blood markers), in the 15-22 week period of gestation if a woman missed the first trimester test.
According to obstetrics guidelines, a pregnant woman is supposed to be clearly told about her screening and testing options, and that includes traditional approaches, NIPT, and even diagnostic invasive testing. Diagnostic invasive testing carries a small but real risk of pregnancy loss, and while the actual numbers of how real this risk is have varied widely in the scientific literature, a historical analysis of this literature suggests that the risk of pregnancy loss from invasive procedures is 0.11-0.22% for amniocentesis and chorionic villi sampling, respectively. This is the primary reason why NIPT has gained so much in popularity, as women don’t want to undergo diagnostic testing if they don’t have to, and place their pregnancy at risk. But because traditional screening is nowhere near as accurate as NIPT, with traditional screening a lot more women end up undergoing confirmatory diagnostic testing that they would not have had to if they took the NIPT test in the first place.
According to a recent massive review of NIPT by the Health Quality Ontario for the use of NIPT in Ontario public healthcare, at best the sensitivity of first trimester combined screening is about 90%, with a specificity of about 95%. For background information on the sensitivity and specificity of the DNA test, please see our previous NIPT post. Basically, this means that with this test, about 10% of women with babies affected by chromosomal abnormalities will never be detected, and about 5% of women tested will be told they have affected fetus when they don’t. That averages to about 1 in 20 pregnant women who take this screening test being told that they should do invasive diagnostic testing to confirm, when they don’t have to because the baby is actually fine. The accuracy of second trimester Quad screening is even worse.
NIPT, on the other hand, based on the Health Quality Ontario analysis of published data, has the sensitivities of 99.5% for trisomy 21 (Down syndrome), 93.1% for trisomy 18 (Edward’s syndrome), and 92.7% for trisomy 13 (Patau syndrome). The pooled specificity for all three trisomies was 99.9%! That means there are far fewer chromosomal problems missed, and at worst 1 in 1000 women are informed of a problem that doesn’t exist, which translates to far fewer invasive testings being assigned. Which was the entire hope and purpose behind the development of the NIPT screening process. The Netherlands, which has adopted NIPT as part of their public healthcare option, has tasked a consortium of scientists to study the clinical utility of NIPT (labelled TRIDENT), and they have been publishing some amazing literature on the topic, based on their thoroughness of investigation. One of their studies suggested that NIPT is likely to miss an existing problem in the fetus only 0.2% of the time.
You can see the value in fully knowing and understanding all of the available options for assessing the health of a pregnancy. While doing diagnostic invasive testing up front would be quite rare to be considered or required, even this option should be explained to expecting parents prior to proceeding with any testing.
According to the Health Quality Ontario report, that is certainly not always the case. In fact, that appears to be the number one criticism voiced by mothers in that a more informed-choice discussion should be taking place. Many women complained that they felt they were never even given a choice, and some, knowing in retrospect what the options are, would even consider never doing any testing! Access to quality information before making a decision is unanimously desired by all mothers, including knowing what the options are, what they test for, and what the potential outcomes are. For many women, the termination of a pregnancy is never an option they would even want to consider, but access to information about the expectations of what might be experienced by giving birth to a baby affected by a genetic condition is very important in order to make the right preparations. Complaints were even voiced that such decisions were often not respected by health-care providers, who tried to coerce women to undergo termination because of the potential severity of the outcome. Ultimately, that decision lies in the mother’s hands.
In the same analysis of patients’ preferences and values, NIPT was very highly regarded. The timing of NIPT was greatly appreciated, being the earliest screening option available (from the ninth week of gestation onwards), especially for women who were tasked with the enormous decision of pregnancy termination which with traditional testing might not be available till much later into the fetus’ development, stages that were already intimately experienced by the mother. The accuracy and non-invasiveness of the screening option makes it far more desirable, confirming the status of pregnancy without the need of going for an invasive option. This sentiment was expressed both by mothers who would elect for termination as well as those who would not, underpinning the emotional value that earlier knowledge and preparation for what is to come can grant. That included mothers of children with trisomies, because it allowed them the time to prepare, and not face the trauma of discovery upon the baby’s moment of entry into this world.
Pregnancy loss – what are the odds?
NIPT was also appreciated for the early preparation for potential pregnancy loss. Chromosomal abnormalities are the leading cause of miscarriage, with an estimated 50% of experienced miscarriages resulting from it. Miscarriage is a deeply emotionally impactful event, and there could be a sense of relief in knowing the actual biological causes behind it as opposed to being left in the dark, where one’s mind might wonder if something wrong might have been done by the mother.
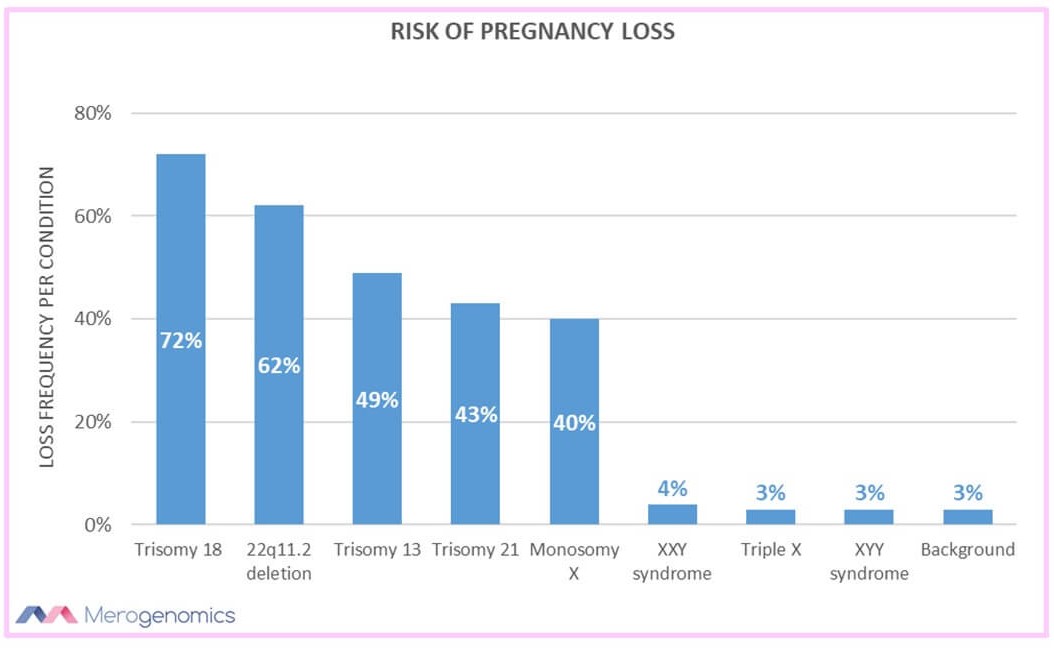
In fact, that was one of the most amazing pieces of information provided by the Health Quality Ontario report, which included the estimated pregnancy loss for different conditions with chromosomal problems. The Trisomy 18 condition leads to the highest likelihood of miscarriages, with 72% of cases resulting in spontaneous miscarriage. The second most common one is one of those conditions affecting only part of a chromosome, termed 22q11.2 deletion syndrome (for the location of chromosome that is affected, also called DiGeorge syndrome), with 62% of cases resulting in a miscarriage.
The chance of having a miscarriage even without any indication is still approximately 3%. Pregnancies are very complex events, and there is a lot of room for different outcomes when we are discussing the intricacy of building an entire human being from a single cell. And this is all based on that code inside your genome.
Which NIPT to choose – full NIPT or not
The final decision that a pregnant woman has to still make, if she elects to go with NIPT testing, is whether she wants to do the common NIPT test that usually just tests for trisomies 21, 18 and 13, or a full NIPT that assesses the status of all chromosomes. The majority of obstetricians that offer NIPT to women, offer the common one that looks at the anomalies of just those three chromosomes. Lately, these tests have been expanded to also include sex chromosomes (those responsible for biological gender classification, XX for females and XY for males), though usually for an additional cost, with some of the anomalies affecting only a fragment of chromosomes. These common trisomies tests were the first ones on the market and have been the most studied.
NIPT tests that asses all of the chromosomes are unique because they can find trisomies with any chromosome. And indeed, trisomies with any chromosome are possible to varying degree. Besides the obvious advantage of being able to catch other trisomies besides the most commonly expected, such tests also appear to have a reduced likelihood of failure, which is still observed at least 2% of the time with the common trisomies NIPT tests. We previously discussed these advantages of the full NIPT test.
One of the cited arguments against the full NIPT test is that overall it has a decreased accuracy, as the sensitivity of the test is not good enough to be able to precisely determine the presence or absence of a given condition because all of these other conditions resulting from an extra or missing chromosome are extremely rare. In other words, the test will falsely inform the pregnant woman of a potential uncommon trisomy that is not even there because the test has a limitation to how precise it is, and the condition is so unlikely to even exist. However, since the implementation of full NIPT tests, there is a continuously mounting body of evidence that all of these other chromosomal anomalies are not that rare!
One study that we had discussed previously showed that the likelihood of any chromosomal anomaly, besides the most common ones, accounted for only 0.3% of pregnancies according to the use of a full NIPT. The authors of that article contrasted that with a 0.6% figure based on chorionic villi sampling. The Netherlands TRIDENT scientific commission have also argued in favour of the full NIPT, observing that other chromosome problems besides those involving chromosome 21,18 and 13 affect 1.6% of all pregnancies! This figure was more frequent than trisomy 13 and almost as frequent as trisomy 18! More than half of these complications were undetectable with a second trimester ultrasound. And nearly all of these events had very severe outcomes for pregnancy, thus proving to be very important information for parents to know and consider with regards to their pregnancy management.
The take-home message with this is that parents should be well informed of their screening and testing options well before it is time to act on these options, so that their path of action can be properly selected. If these choices require DNA testing options, whether prenatal DNA sequencing or full NIPT, Merogenomics can assist with the selection of the best quality choices.
But above all, happy healthy pregnancy outcomes everyone!
This article has been produced by Merogenomics Inc. and edited by Kerri Bryant. Reproduction and reuse of any portion of this content requires Merogenomics Inc. permission and source acknowledgment. It is your responsibility to obtain additional permissions from the third party owners that might be cited by Merogenomics Inc. Merogenomics Inc. disclaims any responsibility for any use you make of content owned by third parties without their permission.
Products and Services Promoted by Merogenomics Inc.
Select target group for DNA testing
Healthy screening |
Undiagnosed diseases |
Cancer |
Prenatal |
Or select popular DNA test
 |
 |
 |
 |
Pharmaco-genetic gene panel |
Non-invasive prenatal screening |
Cancer predisposition gene panel |
Full genome |