COVID-19 vaccine exosomes and interferon cytokine disturbance
Dr.M.Raszek
In this article, we are going to look at one proposed mechanism of how vaccine mRNA could be negatively affecting immune response in certain individuals.
Not long ago, a group of authors published a preprint article (meaning not peer reviewed science article) about possible mechanisms of how spike protein could be leading to different diseases including cancer. Clearly the topic is somewhat inflammatory (pun intended) and has drawn some criticism, but the reason why it has captured Merogenomics attention especially, was because it discussed in detail the role of exosome in the potential development of such diseases. We recently touched on exosome when exploring the unusual claims of women having menstrual cycles affected through mere exposure to vaccinated people, hence the topic of exosomes is of fresh interest.
What are exosomes?
Exosomes are tiny particles of cells if you will, approximately the size of a virus. If you wanted to compare viruses to anything that our body produces, then the closest we could come to are exosomes. In reality, exosomes are like message pods from our cells released into circulation, allowing cells to communicate with one another. These exosomes, made of the same material as our cell membranes, namely fat molecules called lipids, can transport both genetic material in the form of RNA as well proteins on its surface. As all of these components are bioactive, exosomes are recognized to play a role in health and disease, but much yet has to be learned.
To give you an example of how cells can use exosomes for communication, it appears that during a viral infection, infected cells can release large quantities of exosomes to let our immune system to respond to the viral attack. And it appears that similar events take place during the SARS-CoV-2 virus infection. This is the topic we want to break down from that review, especially since exosome production appears to be elevated post vaccination, including the embedding of the spike protein in the membrane of exosomes, and thus the distribution of spike protein throughout the body. This topic was previously covered in one of the Merogenomics videos.
We will be coming back to that specific study shortly.
Another study also investigated the potential role of spike protein in exosomes, proposing that the spike protein can influence the genetic cargo of exosomes with subsequent potentially dangerous immunological consequences at various distant organs. These authors infected cells with genetic instructions to produce the spike protein and isolated and studied the exosomes from such cells. They showed that such exosomes contained specific genetic material in a form of RNA, specifically a type of RNA called micro-RNA. We have previously discussed micro-RNAs involved in the stimulation of the sense of pleasure. In essence, micro-RNAs are super tiny RNA molecules that are used to regulate the use of messenger RNAs, or mRNAs, which are the blueprint instructions for the production of proteins. The mRNA vaccines work in exactly this manner, by injecting blueprint instructions into our cells to produce the spike protein. How injection of such instructions into our body might be influenced by micro-RNAs, or how it might affect our regulatory micro-RNAs is not really known. But we do know that micro-RNAs typically regulate the use of specific mRNAs by binding to them and temporarily preventing their use. Hence micro-RNAs are used to fine-tune how specific proteins are produced in response to environmental stimuli.
Since exosomes are such tiny vehicles for transporting biological signals, it is not surprising that they especially seem to transport micro-RNAs in them. The above authors not only showed that spike-producing cells released exosomes with specific micro-RNAs, but they also showed that these specific exosomes when exposed to microglia cells, which are brain-specific immune cells, microglia happily took up such exosomes and responded to these exosomes with the acute inflammatory response. Acute inflammation initiated by such brain microglial cells is known to be a byproduct of other viral infections such as HIV or Dengue. Hence these authors warned that exosomes could be the responsible factor of the neurological symptoms observed in severe cases of COVID-19.
Then we should ask…
How do micro-RNAs mess with interferon function?
The two micro-RNAs that stood out in the above exosomes from cells injected with genetic material for spike protein production were miR-148a and miR-590. In the brain microglia immune cells, uptake of the exosomes containing these micro-RNAs elicited inhibition of the production of specific proteins: ubiquitin specific peptidase 33 protein (or USP33 protein) and IRF9. Specifically, miR-148a inhibited USP33 protein production by 50%, while miR-590 blocked IRF9 production by 60%. We will get to why that might be so dangerous to promoting acute inflammation right away.
First though, it bears adding that a study of the micro-RNA profile in the blood of COVID-19 patients did not show miR148a and miR-590 to be affected. Thus this observation might be only unique to the type of cells that were being studied, or perhaps only unique when introducing genetic instructions for spike protein into the cells. Meaning, the micro-RNA profile of exosomes in individuals naturally infected by the SARS-CoV-2 virus might be different from those receiving genetic instructions such as the mRNA vaccines. Something we should probably work out in greater detail.
Now let us unpack how inhibition of these proteins could be problematic.
The key word in the IRF9 protein name is interferon (abbreviated IFN). Interferon is a very important cytokine in the innate immune system's fight against viral infections. IRF9 is a signaling molecule that relays the effect of interferon cytokines contacting immune cells, in essence, enact biological reaction to the interferon cytokines.
When interferon cytokine binds to cell surface receptors, it leads to the activation of another protein called JAK. This protein then adds a phosphate molecule to other proteins. Such phosphate attaching proteins are called kinases and there are tons of different types. In essence, it is a way to turn things on and off inside the cell. Without phosphate, a protein will behave one way, but once it has a phosphate molecule attached to it, it will do something else. It is one of the more basic signaling mechanisms used inside cells.
JAK kinase attaches phosphate molecules onto a couple of other proteins called STAT1 and STAT2. At that point, phosphorylated STATs can bind and form a complex with IRF9. This complex is called IFN-stimulated gene factor 3 (ISGF3), which subsequently travels into the cell nucleus where it can lead to the activation of numerous genes involved in anti-viral responses. Or in other words, this ISGF3 complex leads to the production of very specific mRNAs which will then lead to the production of specific proteins that will execute the anti-viral properties attached to interferon cytokines. Thus you can appreciate how important both the interferon cytokines are in fighting virus infection, as well as the ability to produce IRF9, and why inhibiting IRF9 production with micro-RNAs could be so problematic.
How about the USP33 protein then?
USP33 removes ubiquitin from IRF9. Ubiquitin is a small protein that exists in all cells, and when attached to other proteins, it marks them for destruction. Therefore the role of USP33 is to protect IRF9 from degradation. Therefore inhibiting the production of USP33 with micro-RNAs would further lead to compromising levels of available IRF9, essentially blocking the cell’s ability to respond to interferon cytokines.
Will the vaccine studies please show up?
While COVID-19 patients might not have typically increased levels of miR148a and miR-590 micro-RNAs in their exosomes, nevertheless, inborn errors of IRF9 production have been shown to increase the risk of severe COVID-19.
Interferon in general might be a very important mechanism in protecting against COVID-19. It has been shown that patients with compromised ability to produce interferon cytokines were likely to experience more severe COVID-19. Rather such patients exhibit blood viremia (infection of blood with SARS-CoV-2) and overproduction of inflammatory cytokines.
Why this is important is because if the mRNA vaccines could interfere with interferon cytokine signaling in some individuals, this could lead to increased susceptibility to COVID-19 in the first weeks following the first vaccination, before an antibody production due to vaccination has had time to take place. It is plausible that this could indeed take place due to exosomes themselves. In the study discussed in the video presented above, the authors discussed that exosomes with spike proteins were circulating already by day 14 following vaccination while no antibodies to spike proteins were found yet.
This hypothesis might be supported by some publicly available data.
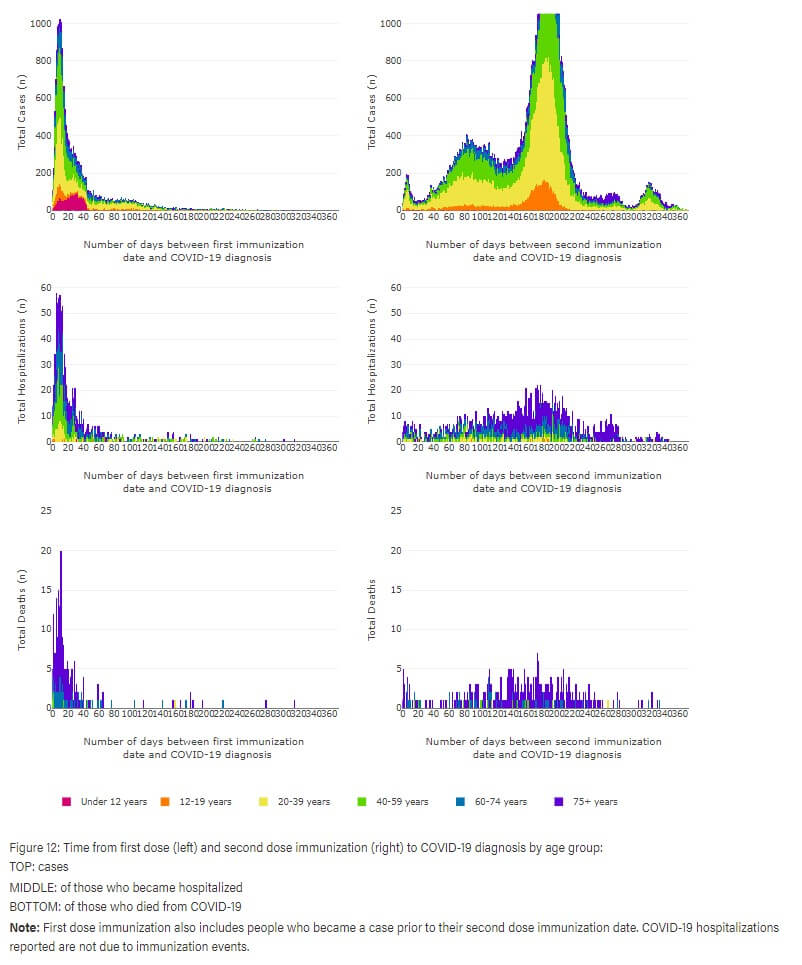
Up to January 14, 2022, the Alberta Health Services used to display the time scale of when vaccinated Albertans would become infected post vaccination. We can retrieve that information from an archived database.
The last archived data can be viewed below:
Vaccination outcomes are shown in the left column post first vaccination and in the right column post second vaccination. We can clearly see in the top row of the left column, corresponding to the observed COVID-19 infection that there is an obvious spike in infections. One could simply attribute it to SARS-CoV-2 exposure prior to antibodies against the spike protein. That definitely could be true, although we also see a similar but much smaller spike post second vaccination as well. Subsequently, in double vaccinated we see the next peak of infection approximately three months post second vaccination, and if we go along with interferon suppression theory, this could be due to waning antibodies that could act on the exosomes featuring the spike protein. In double vaccinated, we then see a third massive peak of infection that likely happens to demonstrate virus immune escape, which dramatically increased susceptibility of the vaccinated population to infection.
In light of the above information, it should be investigated more closely if and how certain individuals might be susceptible to suppressed interferon cytokine production post vaccination. There might be some evidence for mRNA vaccination not inducing interferon response. The reason why this is important is that the use of interferon cytokine is an important aspect of our immune protection, and its perturbation could also affect experience with other infections or health issues dependent on such protection.
The take home message is that biological responses to vaccination could be complex and varied, including possibly in some instances unfavourable consequences if affecting appropriate innate immune responses to viral infections. The authors of the review summarized an interesting molecular pathway that should continue to be investigated to identify potential biomarkers that would allow us in the future to identify high risk individuals of unfavourable response that could then either contemplate the use of a different vaccine.
Molecular biology is complicated and we have yet to learn much about how mRNA vaccines influence such biology within us post injection so that we can continue improving the use of this exciting future technology.
This article has been produced by Merogenomics Inc. Reproduction and reuse of any portion of this content requires Merogenomics Inc. permission and source acknowledgment. It is your responsibility to obtain additional permissions from the third party owners that might be cited by Merogenomics Inc. Merogenomics Inc. disclaims any responsibility for any use you make of content owned by third parties without their permission.
Products and Services Promoted by Merogenomics Inc.
Select target group for DNA testing
Healthy screening |
Undiagnosed diseases |
Cancer |
Prenatal |
Or select popular DNA test
 |
 |
 |
 |
Pharmaco-genetic gene panel |
Non-invasive prenatal screening |
Cancer predisposition gene panel |
Full genome |