How new emerging variants are outsmarting our antibodies
Dr.M.Raszek
Neutralizing antibodies, our cherished friends
There appears to be two concerning forces at play as the SARS-CoV-2 virus continuously mutates: mutations that make the virus more infectious, and mutations that allow the virus to evade recognition by the antibodies that are attacking it. In the case of the latter, this is referred to as immune escape and is currently very worrisome because then a virus could transform itself to a form that will render it immune to the effect of vaccines.
The way that such new emerging variants are tracked is through genomic surveillance, where isolated viral samples are being decoded for their genetic code to see what mutations are present. One of the best such programs is in the UK and we have previously highlighted these efforts in a past video (see below).
One very strong candidate for the evasion of antibodies, including those of vaccines, that you might have heard about, is the Lambda variant (also known as the C.37 lineage). We wanted to drill in a bit deeper here to show you on a molecular level how cleverly the virus has mutated to start escaping the antibodies specifically mounted against the viral spike protein. This variant only recently came on the WHO radar, obtaining the designation of a variant of interest or VOI in June 2021.
First, let’s start by introducing a specific name for a category of antibodies. Neutralizing antibodies. These are the antibodies that effectively prevent the virus from being an infectious agent. Hence the antibodies that neutralize the virus, remove its ability to be a weapon against us.
This should also hint to you that there must also be antibodies that are not neutralizing at all. Meaning these antibodies could bind to the virus but they would not be able to prevent the virus from further infecting cells. Sometimes these antibodies that are not neutralizing can actually be dangerous to us, and we will talk about that as well when we get to the category known as enhancing antibodies.
When we are talking about antibodies that are neutralizing against SARS-CoV-2, we specifically mean antibodies that can bind the proteins exposed on its surface in such a way that the virus will no longer be able to infect cells anymore. Once bound by these antibodies, the virus can meet multiple different fates of destruction, as we outlined in our last post about how a virus is cleared by antibodies.
One important way that antibodies very effectively help to neutralize a virus is by the antibody binding to a viral protein that then blocks or prevents the virus from successfully interacting with our host cells when it tries to gain entry. For a SARS-CoV-2 virus, this interaction with our host cells is mediated by binding of the viral spike protein to our own receptors called ACE2.
You can easily appreciate that any antibodies that prevent the virus from interacting with the ACE2 receptor will be effective neutralizing antibodies. And indeed, many neutralizing antibodies bind to the spike protein at exactly the same location where the ACE2 receptor would bind. Neutralizing antibodies could also bind away from the spike protein motif recognized by ACE2 receptor and still prevent successful interaction.
Where antibodies actually end up binding to the viral proteins can be a unique biological process in each person. Hence each person creates their own version of effective antibody defenses, and the quality of these can range widely between people. We brought up examples of this in our article about how genetics could predispose a person to COVID-19.
But the point is that each person uniquely creates their own army of antibodies of varying quality against a virus. Within that army can be differing levels of effective neutralizing antibodies.
Let’s bring back the Lambda variant into this.
Lambda variant is using wild tricks
The reason why Lambda is a variant of interest that is suspected of having an enhanced evasion of neutralizing antibodies is because Lambda variant has a dramatic deletion in its viral genome that results in a segment of the spike protein that was supposed to be built but instead is completely absent. In other words, this deletion in the viral genetic code results in a corresponding deletion in the spike protein itself. It is this deletion of a fragment of the spike protein that changes enough of the overall three-dimensional shape of the viral spike protein, so that this segment is simply no longer recognized by certain antibodies that were binding this region. It appears that this particular region of the spike protein was vulnerable to neutralizing antibodies, and a virus found a viable mutation with the large effect of evading antibodies without appearing to compromise anything else as a protein.
This Lambda variant deletion is a unique 7-amino-acid deletion mutation (amino acids are the building blocks of proteins), RSYLTPGD246-253N (where 8 amino acids are substituted for a new single one, with each amino acid designated by a capital letter here). This region of the spike protein that is normally vulnerable to neutralizing antibodies but that is shifted in shape in the Lambda variant is referred to as the N-terminal domain of the spike protein.
Think of a singular subunit of the spike protein as a torso with two heads. Remember, final spike protein is actually made up of three identical spike proteins coming together to form a final structure we observe on the surface of the virus. But if you just take one of those three identical units, this is what would look like a torso with two heads. One head is the receptor binding domain that is used by the spike protein to bind to ACE2 receptors on the surface of our cells. The second head is the N-terminal domain, where in a final conformation of the spike protein, the N-terminal domain closely hugs the ACE2 receptor binding domain of the adjacent spike protein unit.
These deletion mutations affecting the N-terminal domain nicely show you the plasticity of viral genetic code and how rapidly it can evolve under favourable conditions (i.e., a rapid life cycle with billions of progeny and many available hosts for SARS-CoV-2). Random mutations can become nature’s most unusual creations. It is simply a mathematical matter of enough random chances being available for the SARS-CoV-2 virus to experience an unusual mutation that could be favourable for survival.
Interestingly another article we discuss in this blog estimates the SARS-CoV-2 mutation rate is approximately 25 new mutations per year.
And genetic code deletions are not infrequent and not the only events in the random mutagenesis that is naturally experienced by any self-dividing structures like viruses and cells. You can also have insertions. Recall that the very unusual presence of a furin cleavage site in SARS-CoV-2 virus is due to such an insertion in this virus. This fortuitous insertion for the virus ends up helping the virus fuse with host cells, and therefore aids in cell infection.
So how serious is this Lambda variant? It looks pretty serious, and another recent preprint study indeed showed that Lambda escapes neutralizing antibodies. While Lambda has other mutations that increase its infectiousness (like, not surprising, the L452Q mutation which affects the area of ACE2 recognition – by the way, Delta variant has a L452R mutation in this same site), it is this unusual deletion and subsequent escape from neutralizing antibodies that is suspected in the spread of Lambda in South America (such as Peru, Chile, Argentina, and Ecuador, in some regions among a high vaccination background). This variant is serious enough that it is monitored on a global scale now, but has not yet been raised to a status of a variant of concern. Luckily, while a serious threat, Lambda has not gained significant dominance yet in the world.
In fact, all major variants appear to escape some neutralizing antibodies, including the Delta variant, and we are talking about neutralizing antibodies produced by mRNA vaccines. Compared to the original variant against which the mRNA Pfizer/BioNTech was produced, Delta variant appears to be 6-fold less sensitive to those same vaccine induced neutralizing antibodies. These are some of the features that make the Delta so particularly dominant right now, including its still high infection rate among vaccinated individuals.
And ironically it might be due to the global dominance of the Delta variant that has stalled the spread of Lambda as thus far Delta appears to be more infectious, even if it does not escape neutralizing effect of antibodies to the same degree that the Lambda variant could.
It is as if the Delta variant for the moment was protecting the population from the Lambda variant because the Delta variant is more effective in spreading through the population.
But Lambda might be a problem in another way.
Antibodies enhancing the virus infection
Remember how we mentioned earlier that some antibodies that are not the neutralizing type can be dangerous?
There are also antibodies that, in theory, by accident could actually enhance the infectivity of the virus. In fact, this is believed to have been observed very early in a pandemic and we previously wrote about how antibodies can promote infection.
Typically, when antibodies that enhance infection are considered, this phenomenon is referred to as antibody dependent enhancement or ADE. But previously, antibody dependent enhancement was thought to occur by promoting the infection of the immune cells that function to recognize antibodies bound to a virus via the stem of these antibodies. This stem, or a stalk if you will, is labelled as Fc in antibodies. These cells are meant to recognize the Fc portion of antibodies binding to the virus (by binding to the Fc portion via a specific Fc-binding receptor), and then internalize (literally engulf) these antibody-virus complexes to destroy them. But if these virus complexes that are internalized by these killer cells somehow get loose inside these immune cells, the killer cells themselves become infected! We wrote about this type of antibody dependent enhancement in our last blog post as well. Therefore, Fc-receptor-mediated antibody dependent enhancement has traditionally been thought to occur only through the infection of Fc-receptor-expressing cells (such as monocytes or macrophages).
But in the case of COVID-19, the antibody dependent enhancement of the infection occurs in a different approach to the traditional one described just above, and has nothing to do with binding of the Fc portion of virus-bound antibodies.
It turns out the N-terminal domain, if mutated in one way, can help the virus escape neutralizing antibodies, and in another way other mutations in this same domain appear to induce the viral infection rate. In the above mentioned early pandemic example, COVID-19 patients produced infection enhancing antibodies because the antibodies captured the spike protein in a state more favourable to bind the ACE2 receptor! This state is referred to as open conformation of spike protein, where the ACE2 receptor-binding motif is made to stick out more and therefore available for recognition by the ACE2. This seemed like an unexpected finding, and it shows you how antibodies can accidentally work against you. The enhancing antibodies in these patients were detected at similar levels as neutralizing antibodies, showing at least one example of how diverse the response of antibodies can be, at least in naturally infected COVID-19 patients.
The same 7-amino acid deletion of the Lambda variant was shown to enhance virus infection of cells by the Lambda variant when bound by an antibody already known to promote infection.
Therefore, this single Lambda variant mutation could be a double whammy! It can help evade neutralizing antibodies and enhance interactions with infection promoting antibodies.
Either way, one or both of these effects might have contributed to the Lambda variant spread thus far and that is a serious concern for the future of the virus, and its potential immune escape from neutralizing antibodies.
Currently, although there appears to be no evidence that vaccines could be promoting the production of such COVID-19 disease enhancing antibodies we just don’t know if anyone has even assessed it yet.
Perhaps there could be one suspicion that it might be happening based on a recent characterization of Delta variant spike proteins, which appear to have mutations that would promote infection if targeted by such enhancing antibodies.
It appears that Delta variant might also harbour mutations in its N-terminal domain that could promote increased infection. In one particular analysis, computer modelling was used to show that Delta mutations could promote infection with use of antibodies in yet another unusual way. Modelling suggested that previously COVID-19 patient-isolated infection enhancing antibody would have even higher affinity against the Delta variant than the original strain of SARS-CoV-2 spike protein.
How does this particular antibody promote infection?
It looks like binding of the antibody to the N-terminal domain of the spike protein helps the spike protein to make contact in this same area with a lipid layer. In nature, presumably this antibody-virus complex would enhance its interaction with the cell surface lipid layer.
In other words, according to this computer modelling, the antibody-virus complex makes a further complex with lipid layer for a final antibody-virus-lipid layer complex. This is the first time we are hearing of such suggestion, so we wanted to show you how the authors demonstrated it with their model as below. The trimer of the spike protein is colorfully shown with the yellow, purple and cyan units. The infection enhancing antibody is shown in green, binding to the one of three possible N-terminal domains. The lipid layer binding to the same region of N-terminal domain is shown in red. In this computer model you can also see that the lipid layer is also making some contact with the antibody, and it is this guidance of antibody to the lipid layer that authors believe could help promote infection by this particular antibody.
These are the type of sneaky events that can take you by surprise and who knows how many more surprises we are yet to encounter as we learn about the different ways antibodies can interact with a virus and tamper with an infection in both inhibitory and stimulating fashion.
This is yet another unconventional form of antibody dependent enhancement, the authors referring to it as a new type of antibody dependent enhancement that is independent of Fc-receptor binding cells. Whether it turns out to even be true, time will have to tell, as none of this is currently verified by real-life evidence. Modelling can be at times a very far cry from reality. And to be honest, it was surprising to see a lipid layer interaction here. We thought it would be quite some distance away considering that this apex of the spike protein that interacts with the ACE2 receptor that itself will be some physical distance still from the lipid layer of the host cell membrane. Maybe the spike protein really is going for a dunk into lipid bilayer of the cell membrane. Maybe that helps to bring the virus closer and to actually fuse with the cell membrane to enter the cell. For now, we will just go with this model as quite interesting.
But it did not stop the authors of this article from going as far as to warn of the possible consequences of vaccination potentially enhancing the infection of the Delta variant through promoting infections via this unusual mechanism. The authors warned that future vaccine developments should consider these sites to reduce the likelihood of enhancing the disease through the process of vaccination.
The same authors also discussed that in their models, mutations in the same N-terminal domain also impact the binding site of previously established neutralizing antibodies, once again, apparently contributing to immune escape.
By the way, while all of this might be new news to you, in the science world it is not. This concept of antibody dependent enhancement is understood well enough that even Dr. Antony Fauci has been warning against its possibility as an outcome of vaccination that we need to be vigilant against. Below you hear Dr. Fauci explain antibody dependant enhancement, stating “It's the worst possible thing you can do...is vaccinate somebody to prevent infection and actually make them worse." It is just that in the zeal of promoting the vaccines, any risks associated with vaccines and their potential for promoting disease has been skipped from discussion, especially since the great original effectiveness of the vaccines argued against any ideas of antibody dependent enhancement.
And do you think this is the only antibody concern we have? Promoting virus infection?
Not even close!
What we are trying to say is to always remember that antibodies are a double-edged sword. They can clear the infection. But it is also immune system dysregulation, including antibody dysregulation, that ends up making the COVID-19 disease outcome more dangerous for severe cases.
Self targeting antibodies
The big original marker of how induced antibodies can be treacherous as well, is once again the study of observed outcomes in surviving COVID-19 patients, and this also demonstrates another potential issue with antibodies. Some severe COVID-19 patients showed increased rates of autoantibodies, or antibodies that targets our own self biological molecules as opposed to the virus. For example, antibodies against our own interferon cytokine, an important self-defence anti-viral agent we wrote about in last blog post.
In one study that looked into this, COVID-19 patient antibodies were incubated against nearly 3000 of our own self targets. The COVID-19 patients antibodies were shown to pursue many internal targets, especially targets in the immune system, but also against specific tissue markers as well. Overall, the take home message by these authors is that this internal self-targeting by antibodies contributes to the disease development as well. The antibody responses to SARS-CoV-2 infection can thus be extremely diverse and complex in their outcomes. At least some of this complexity and diversity of responses should probably be also expected as an outcome of vaccination.
The long-term consequences of antibodies created to fight an infectious pathogen, which can exist for long period of time, on potential self attack is probably yet to be understood. We do not even know if it is possible that part of virus mutagenesis is precisely to affect the long-term effectiveness of antibodies against the virus that they were supposed to destroy, and to instead actually help preserve the virus. Could some of the antibodies developed against the virus actually be used to tweak other immune system elements targeting the virus? Could the evolutionary cat and mouse game between the virus and host extend that far? This does really happen, when antibodies generated against a pathogen also attack the host, and is referred to as pathogenic priming. It is suspected that pathogenic priming could play a role in COVID-19.
Future developments?
And Lambda variant is not the only variant with which we need to be concerned. By now, it really looks like the evolution of the virus is heading in the direction of bypassing as many neutralizing antibodies as possible. You will see that right away, in fact.
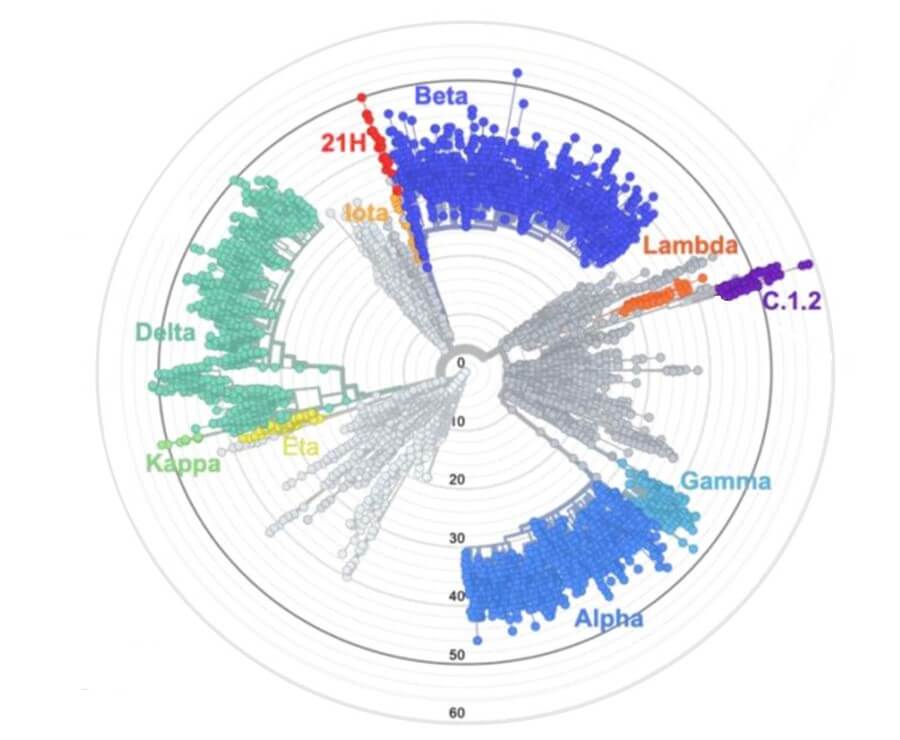
Another variant of potential concern is the newly emergent variant from South Africa, the C.1.2 variant which also happens to be actually the most closely related variant to the Lambda variant. Just to give you the notion of how different this virus is now, the C.1.2 variant harbours somewhere between 44 to 59 mutations that are not present in original Wuhan virus strain that started the pandemic which would rank it as one of the most mutated strains thus far. One of the most interesting graphics presented by the authors in the study of this new variant was a phylogenetic map of the different SARS-CoV-2 variants as they emerged, indicating how many mutations it took in comparison to the original starting virus strain. You can see that all of the variants of concern and variants of interest thus far are quite heavily mutated compared to what has initially started the pandemic. The closer any points are towards each other on this map, the closer they are genetically related, so this map also shows you how distantly or closely related different strains are to one another. Now you can see how related the different strains you hear about are to one another. It is clear the mutational potential of the virus is enormous. And the C.1.2 strain is the most mutated yet.
At least 14 of these mutations in the C.1.2 strain are in the spike protein of SARS-CoV-2. Authors suspect that this variant arose in a single individual that was infected for a prolonged period of time, like some events we have discussed in our past posts as well. Many of the mutations have been seen in other variants already, indicating a convergent evolution of the virus towards a form that is more infectious, and that is more capable of evading neutralization by antibodies. In the case of C.1.2 strain this immune escape could be promoted in particular by the deletion mutations Y144del, 242-244del (notice, right next to the Lambda deletion described above! This is fast becoming the virus’ evolutionary favourite trick!), as well as the E484K mutation. While the impact of antibodies against this variant are currently being assessed, the worry here is that the frequency of this particular variant in South Africa is slowly increasing, and if indeed this variant will turn out to bypass antibodies, this type of strain is not something we want to see emerge on the global scene.
The take home message is that evolutionary forces are shaping the virus development with two potential negative outcomes to our mass vaccination program: allowing the virus to escape the effect of neutralizing antibodies, and promoting virus interactions with antibodies that actually further promote the infectivity of the virus as opposed to neutralizing the virus. Based on actual antibodies collected from COVID-19 patients, it appears that both types of antibodies can be produced as a consequence of exposure to SARS-CoV-2, the neutralizing antibodies and enhancing antibodies.
Could the antibodies that enhance the disease progression be produced as a consequence of vaccination in some individuals? Currently no one appears to have demonstrated that, but it remains a possibility, and based on antibodies found naturally in patients, we should expect to find it eventually in some rare individual instances after vaccination. As already mentioned above, some scientists have proposed this mechanism as a potential reason for the observed high infectivity of vaccinated people by the Delta variant, although that remains a mere hypothesis. In the end, the effectiveness of the vaccination campaign might be determined by the mutational evolution of the virus to escape the neutralizing antibodies (what we are observing) and perhaps promote interaction with enhancing antibodies if such enhancing antibodies are indeed produced post vaccination (no evidence). In those COVID-19 patients, it was the balance between neutralizing versus enhancing antibodies that appeared to be the contributing factor towards the disease’s severity progression, while luckily for most people, SARS-CoV-2 infection is not a threat to develop the severe form of the COVID disease. But we are now slowly learning, that there can be antibodies produced that will not be helpful to the host, and what we are observing is that the virus mutagenesis is appearing to take advantage of that biological phenomenon.
Who needs to be most aware? We still do not fully know.
What are we to do now?
One of the Lambda preprints discussed above concluded, in the context of vaccines that still allow spread of virus infection, what makes good sense is to do this: “massive vaccination campaigns in countries with high SARS-CoV-2 circulation must be accompanied by strict genomic surveillance allowing the identification of new isolates”.
In other words, we need to be vigilant in monitoring virus mutagenesis to avoid the potential catastrophe of virus immune escape that could render vaccination (and its hard-won benefits of reducing the negative impact of the COVID-19 disease) ineffectual.
Especially since the mutation rate of the virus might be increasing! In the above-mentioned C.1.2 strain, the authors estimated the mutation rate of the C.1.2 variant to be 1.7 faster than the current global rate of SARS-CoV-2 mutagenesis. At this rate, it is just a matter of time scenario before we see further escape from antibody neutralization.
Thus, mass screening of the population for infection with rapid assays, followed by careful tracking of the developing viral strains is one of our best weapons right now. The more careful and diligent we are in this approach, the more likely we are to perhaps avoid the negative consequences of the virus evading neutralizing antibodies.
This article has been produced by Merogenomics Inc. and edited by Jason Chouinard, B.Sc. Reproduction and reuse of any portion of this content requires Merogenomics Inc. permission and source acknowledgment. It is your responsibility to obtain additional permissions from the third party owners that might be cited by Merogenomics Inc. Merogenomics Inc. disclaims any responsibility for any use you make of content owned by third parties without their permission.
Products and Services Promoted by Merogenomics Inc.
Select target group for DNA testing
Healthy screening |
Undiagnosed diseases |
Cancer |
Prenatal |
Or select popular DNA test
 |
 |
 |
 |
Pharmaco-genetic gene panel |
Non-invasive prenatal screening |
Cancer predisposition gene panel |
Full genome |