COVID-19 PCR tests – are we over-testing or under-testing? Who is right?
Dr.M.Raszek
COVID-19 testing – a polar divide!
There is a massive public debate going on about the COVID-19 pandemic threat and whether it merits the draconian measures enacted against the population. Wherever you might stand on the spectrum of this debate - whether you decry anytime someone dares to step outside into the fresh air, or you belittle the existence of this infection as nothing more significant than a bad flu – there is one argument used by either side that gets a lot of collateral damage: the use of PCR tests to diagnose the infection with the SARS-CoV-2 virus. Those subscribing to the religion of full-lockdown measures to protect our innocents from certain death, propose that we should test everyone on a regular basis, and only those with negative results, i.e. not infected with the virus, should be allowed to step outside their domestic prison. Whereas, those pandering to the pandemic as a “plandemic” religion , ridicule the PCR diagnostic test as highly inaccurate, resulting in ridiculously inflated numbers that do not resemble even the image of reality, and should never be used as a justification for such enormous limitations on personal liberties; destruction of our economy; and even to induce unintended mortality associated with public lockdowns by suicides, limited access to first responders, or reduced access to those necessary health-related facilities by the most vulnerable members of our society.
So where do we really stand on these PCR tests? Which side might be right and which side is wrong?
The answer is neither! Both sides are wrong and this stems from a poor understanding of the science behind these PCR tests! Although somewhere along the way PCR tests began to be used differently , without the proper scientific and statistical context, and one side can now have stronger claims to support their conviction. But that all really depends on one massive question: how common is the infection in the population?
What is PCR test? You are going to get this (bad pun intended)!
Before we get into explaining our logic, let us give a “small” background on PCR. PCR stands for Polymerase Chain Reaction. Sounds really fancy. And it is! It uses a smart utility of nature’s biology in a technical assay. At the heart of this is a protein called polymerase. A polymerase is a protein (we compare it to a molecular robot) that copies DNA inside cells. It is a necessary part of living systems in order for cells to be able to duplicate their genetic material before dividing into two daughter cells. So that process is our star here. Where does the “chain reaction” come from? From our manipulating the system so that our star polymerase can copy the same fragment of DNA we want over and over, but with each round of copying, you obviously double the amount of DNA to be copied in the next round. This allows you to copy a miniscule amount of DNA rapidly into a huge amount!
Here is an old but awesome and short video explaining this simple concept.
In this video you are introduced to primers, or tiny fragments of a single stranded DNA that can bond to other single stranded DNA, provided that the sequence between these two single stranded DNAs is complimentary. Recall that DNA is composed of four chemicals called nucleotide bases that are abbreviated as A, C, T and G, and these chemicals are complimentary - meaning that nucleotide A has an affinity to bond to nucleotide T, while nucleotide C is especially fond of interacting with nucleotide G. This allows DNA to exist as a double stranded helix where two complimentary sequences of single stranded DNA can interact with one another . This also makes it super easy (and fun) to copy DNA as when you copy either of the single strands, you will end up with two identical DNA double strands! And by the way, it is the arrangement of just these four chemicals in a very specific order that is all of the biological information that is your book of life – your genome! It determined how you came into existence from a single fertilized cell to the incredibly complex multicellular individual that you are, and thus it is the blueprint of how you exist from day to day. Or be severely compromised if this code is mutated, and/or even cease to exist if this code is really badly broken!
In PCR we can take advantage of this DNA complementarity to define the exact region of DNA we want to copy to infinity (amplification), just like you saw in the video above.
But in case of the SARS-CoV-2 virus, its genetic material is not DNA, but rather RNA. For a review of relationship between DNA and RNA, look at our last ASHG2020 related blog post. Luckily, we have polymerases that can copy RNA into DNA; these cool little robots are called reverse-transcription polymerases. Transcription is a word scientists use to describe copying of DNA into RNA. That might not sound very intuitive so they really tried to make up for it with coining the phrase “reverse-transcription”. Thus, the PCR tests we use to copy RNA are RT-PCR.
If you want to get technical, the PCR test for the detection of SARS-CoV-2 virus is RT-PCR. You might have also seen qRT-PCR. The q stands for quantitative, which means that the virus quantity in the sample can be measured, but such tests require special controls of known amount of material to be run. That is like a fancy-schmancy test that helps to determine the actual viral load in the sample. Let us expand on that here because this will be important later on. Recall that PCR amplifies genetic material by copying it over and over in rounds of cycles. And each cycle you double the amount available DNA to be copied in the next round. PCR tests give you a result that includes a minimum cycle number which was required to pass a certain threshold of detection. It is referred to as Ct or Cycle threshold. Basically, the lower the Ct value, the more starting virus genetic material was in the sample (higher viral load) and therefore the faster the confirmatory detection threshold was crossed. The higher the Ct value, the less virus genetic material was present in the sample (lower virus load) and the more cycles it required to detect the virus in the sample. The example of COVID-19 Ct values provided by the Public Health Ontario is Ct ≤ 38 would be positive and Ct > 40 would be negative. The in between is a gray-zone that would require retesting.
Unfortunately, Ct values between different tests cannot be compared unless you know also how much starting material is used. If that starting material is different, it can lead to slightly different values. Another caveat is that other factors could also be affecting the Ct value, besides the amount of virus genetic material in the sample - so the measure of viral load this way might not be precise. Hence the PCR results are typically used for confirmation of a diagnosis, and not to actually determine the quantity of the viral load.
Before we move on, the point is that RT-PCR test allows you to amplify very specific regions of your choice in the SARS-CoV-2 genome. This will be important for further discussion. The second very important point is that PCR identifies presence of SARS-CoV-2 RNA only! If you get a positive PCR test result, it means that the test identified the presence of RNA genetic fragments belonging to SARS-CoV-2 virus in that sample (usually from those enormously popular “poke-your-brain” swabs). This will also be important for further discussion. If you get a negative PCR test result, it means that no SARS-CoV-2 genetic material was found in the analyzed sample.
COVID-19 PCR test false positives
With that handy background, let’s now revert back to the argument that PCR tests are not accurate. The zealots who demonize the PCR-everyone-in-this-world ideology love to bring up the fact that PCR tests have high rates of false positives, and/or they say that it is not sensitive enough. But that is all depending on how you define these terms! A false positive just means that the PCR identified the presence of the SARS-CoV-2 virus when the person was actually ok. Meaning, they never got sick because their body was able to handle the biological insult, and they were never infectious.
And here is the rub! It all comes down to details. Remember, we call these PCR tests diagnostic. But “diagnostic test” is supposed to have a very specific meaning to it. Meaning it is supposed to diagnose a specific condition. In the case of these PCR tests, the goal would be to diagnose COVID-19 condition. The identification of presence of SARS-CoV-2 virus itself in a sample is not diagnostic of any condition. It simply means there is a presence of a virus in the sample. We assume that it automatically means that you have COVID-19 condition, but that is not necessarily the case.
The important issue here depends on how we define diagnostic accuracy. Do we define it by the presence of SARS-CoV-2 or by having clinical COVID-19 conditions?
Let us look at the accuracy of the presence of SARS-CoV-2 first. This is referred to as analytical performance of the test.
In this regard, in fact, the test is extremely accurate! Remember that PCR allows you to amplify very specific genetic regions of interest. Therefore, you can select a genetic region of SARS-CoV-2 that is unique to SARS-CoV-2, and not present in other viruses. Furthermore, to decrease the likelihood that the PCR test will not be identifying any other related virus besides SARS-CoV-2, often more than one genetic region of SARS-CoV-2 is inspected at the same time in the test. The World Health Organization (WHO) currently lists seven qRT-PCR tests using different SARS-CoV-2 genetic targets, all developed by authoritative health bodies all around the world. The DNA primers used for designating which regions of SARS-CoV-2 are amplified would be screened against the genetic information of other viruses, the human genome and bacterial genomes to ensure there is no possibility of amplifying such non-intended targets. The amplification of non-intended targets is referred to as cross-reactivity.
But the “plandemic” proponents like to say that these PCR tests pick up other viruses, like the common cold viruses which are also coronaviruses, and hence we are getting soooooooo many positive cases which are actually not true positives. This could potentially be true if the PCR test result showed a really high Ct value as that is when the cross-reactivity threat comes in because if there is no SARS-CoV-2 virus in the sample, those test DNA primers are constantly searching for some genetic material to bind to and for the polymerase to then amplify that material. Sometimes a non-perfect match can occur between the primer and its target, and something other than SARS-CoV-2 virus is actually amplified, leading to a false positive result.
How likely is it that the PCR test for SARS-CoV-2 could identify other viruses?
Let’s take a look.
The Public Health Ontario claims its rate of COVID-19 false positives, i.e. identifying presence of the SARS-CoV-2 in individuals that were not infected, based on more than 850,000 samples tested, was less than 0.01%. Basically, non-substantial. Which is incredible!
Their explanation behind reasons for false positive rates does not even mention cross-reactivity explicitly. It is not even on the radar. They divided it into the following categories:
- Pre-analytical errors – errors prior to the actual testing being done which could include samples being mislabelled, or accidental sample contamination either in transport or in the laboratory
- Analytical errors – errors during the actual laboratory testing, which could include contamination of test reagents or defects in the equipment or reagents used
- Post-analytical errors – errors in results interpretation by the technologist or accidental errors in reporting of results
Although looking at past historical records, such low rate of false positive rates with the PCR is typically never observed! One recent analysis of such past PCR detection performance, with approximately 10,000 combined negative tests for various viruses such as influenza, original SARS, Zika or Ebola, together yielded approximately 3% false positive rate. The same recent analysis looked at all the published evidence of analyzing false positives of SARS-CoV-2 (surprisingly extremely scant data for the fact that we are using this test on millions of people! That is kind of ridiculous) and they saw a range from 0.3-3%, so more in touch with reality than what was claimed by the amazing efforts of Public Health Ontario laboratories. These numbers will be very important for discussion below.
But the highest likelihood of false positives mentioned above is from human error, and not in the performance of the test. In terms of potential cross-reactivity, one of the most authoritative publications we found on this topic was a study that compared the performance of COVID-19 PCR tests developed by four different national public health institutes or universities, the China Center for Disease Control, United States CDC, Charité Institute of Virology of the Universitätsmedizin Berlin and Hong Kong University. When using samples collected from patients with respiratory problems prior to the COVID-19 outbreak, there was no cross-reactivity observed. That does not mean that this issue was always so rosy. Indeed, early on in the pandemic, the US CDC had to withdraw one of the early proposed PCR tests from use precisely due to cross-reactivity.
What about the super closely related viruses like the original SARS or related coronaviruses viruses from bats? Yes, in theory these viruses could indeed be picked up due to their very high similarity to SARS-CoV-2 although these are not circulating in the population and therefore they are not going to be an issue. We are not going to be misdiagnosing due to these viruses.
What does it mean to diagnose COVID-19?
Now let’s look back at diagnosing COVID-19 with the PCR test. This is referred to as clinical performance of the test (in contrast to the above-mentioned analytical performance). COVID-19 condition means being infected with the SARS-CoV-2 virus that also makes you contagious to others (for a specific window of time), and puts you at a risk of developing associated symptoms, the most acute of which can require hospitalization and even time in the intense care unit. The most extreme end, of course, is death itself.
Therefore, if we define the PCR test to be a diagnostic for COVID-19, a condition that is expected to have certain clinical symptoms, then people who have tested positive are highly expected to develop COVID-19 and thus require special attention (monitoring, isolation, treatment). Indeed, the PCR tests that are used on everybody were originally designed for the purpose of being diagnostic for COVID-19.
What this means is that the PCR tests were originally designed for testing a very specific group of people only at high-risk of actually having COVID-19. That would include symptomatic people, i.e. those coming to clinics and hospitals with symptoms that could be suspicious of COVID-19 (but could also be symptoms of other unrelated respiratory infection) and confirming whether the condition that such patient has is COVID-19 or not. It could also include individuals with previous medical history of COVID-19 or individuals with presence of antibodies to SARS-CoV-2 virus, and finally people who might have had exposure to COVID-19. In other words, the COVID-19 diagnostic PCR test is for people with a high pre-test probability of the disease.
Therefore if we look at the sensitivity of the PCR test to strictly confirm the diagnosis of COVID-19 condition, and not merely the presence of the virus itself, the sensitivity is not as high. The detection rate can even drop to ridiculously low levels, depending how long after the symptoms presented that the test was performed, and this all has to do with how much virus will be available in the sample. The highest PCR test diagnostic performance is within the first days of COVID-19 symptoms onset. And if a sample collection does not capture the virus in an infected person due to the sampling method itself, it is definitely possible to see negative results followed by subsequent positive results. This is because as we stated above, the sensitivity to detect SARS-CoV-2 is extremely high. But the method of sample collection does not guarantee that virus will be captured necessarily. Likewise, some discrepancies like these can exist depending from where in the body the sample was collected. As a consequence, the issue is that the diagnostic PCR test results in many false negative cases, meaning failure to diagnose COVID-19 patient on the first test.
One meta-analysis of many studies already published, together including over 12,000 confirmed COVID-19 patients indicated overall false negative rate of nearly 9%, but ranging from just under 2% to nearly 33% (for the gold-standard nasopharyngeal samples, those poke-your-brain samples)! All cases were confirmed with repeat testing. So the sensitivity of the diagnostic COVID-19 test can range from 70% to nearly 100%, depending on the study. Authors pointed out that the quality of published data was low, and this report was a preprint (see our previous post for definition).
But that is not the main issue! The test is still diagnostic option we have for people at high risk.
The main issue is that the PCR tests were not designed to be used for diagnostic purposes in asymptomatic people with no exposure to COVID-19! Or in other words, the COVID-19 test was not designed for people with a low probability of disease.
When testing asymptomatic patients, a negative test does not rule out infection, as it could be early in the incubation period before the virus is actively replicating at a level that can be detected by PCR assays. But when testing asymptomatic patients, a positive test does not rule in infection either.
In fact, we do not truly know what the sensitivity, specificity, and predictive values of testing in low-risk asymptomatic people are. For us to truly know that, we would need to test large number of patients with clinical follow up and repeat retesting at intervals to truly determine the diagnostic power of the PCR test in asymptomatic people.
The moment we switched to testing asymptomatic people with a low probability of the disease using a diagnostic PCR test, it is in fact no longer a diagnostic test, but rather a screening test. This means that the current screening strategy we are doing on the population is not good enough to truly determine if someone with positive result will indeed end up with COVID-19 condition, and such positives would have to be followed up with diagnostic test for COVID-19. Which ironically is also the same PCR test, but only for those who already have symptoms or are in high-risk of having a disease.
So what does it mean if an asymptomatic, low-risk person receives a positive PCR test result? It could mean that the person will go on to develop COVID-19 and will be contagious at some point along the way. But it also could mean they never will, and it will have absolutely no bearing on their health, and they will not be contagious at all and will not be any threat to public safety! In that case that is a totally false positive!
Why could many asymptomatic cases be false positive for COVID-19 test?
Remember that the diagnostic PCR test is designed to look at the genetic information of the SARS-CoV-2 virus. This means it will detect the genetic material irrespective of its source in the body, whether it was intact and still infectious sort of virus, or already broken-down virus with its genetic material exposed for capture. In essence, the PCR test does not test directly for intact functional virus particles. Thus, while we could be detecting a virus in a person for weeks after infection, this might not at all correlate to that person being contagious at all. This could even mean detection of virus in a person that was never ill and never infectious because they did not have sufficient virus amount in their body to get to that state! The take home message is that PCR test does not distinguish between infectious and non-infectious virus
The only way we can ascertain that correlation is by taking such samples that produced positive test results and then attempt to culture an intact virus from this sample. We can then know if an intact virus was indeed present in the sample if it continues to infect and kill cells in a laboratory test.
Such studies have indeed been done and continue to be done. Some such studies even correlated their finding to the PCR test result’s Ct values. The PCR samples that did indeed have infectious viruses were dependant on the amount of virus detected by the PCR in the test. This is referred to as viral load. In other words, for a person to be considered contagious, a certain minimum amount of virus has to be present in that sample. Below that certain amount, such samples failed to infect and kill cells in laboratory tests.
Let’s see what was observed in one such study of symptomatic cases. The ability to culture the virus from the COVID-19 samples of these individuals peaked around the time of symptom onset with a median duration of virus shedding of 4 days. The ability to culture the virus in the first week post symptoms onset was 74% and this declined to 6% 10 days after symptoms onset. For Ct values of ~23 the virus could be cultured nearly 87% of the time. For Ct >35, the ability to culture the virus dropped to 8%.
That was a recent study. Very similar results were seen early on in the outbreak of the pandemic. COVID-19 patients with Ct ≥ 34 had no detectable viral culture, and therefore were presumed to no longer be able to excrete infectious viral particles. No virus was cultured after 8 days post symptoms. This is potentially crucial because the virus was still detected with PCR for up to 21 days after onset of symptoms! That is very wide window of detection with PCR without ability to see infectious virus. The treatment institute behind these observations even proposed that patients with Ct ≥ 34 values should be discharged from the hospital.
These studies of correlating PCR test result and Ct values was even done for surface contamination! Ct values of ~29 correlated with the detection of culturable virus, whereas Ct values of ~33 correlated with the detection of non-culturable virus.
You probably see a pattern emerge here. While Ct values cannot be compared directly, high Ct values suggest that no infectious agent might be present in the sample at all! But currently it is still a COVID-19 diagnosis. US CDC also commented on similar findings of their own, as did Canada’s National Microbiology Laboratory (except they did not observe the ability to culture viruses at Ct >24, and also not past 8 days from the onset of symptoms), and we stopped looking after that.
The point is that health authorities are fully aware of this. This is not news. This is why health authorities actually discouraged using a diagnostic PCR testing as a screening tool the way we started doing it. Thus Public Health Ontario does not recommend COVID-19 asymptomatic testing, as does the US CDC. It is well understood the significant negative impact that tests with high false positive rate can have, be it financial, psychological and societal due to the misdirection and misuse of resources. Our biggest risk of using diagnostic PCR tests for screening purposes is that we are basically running the risk of inaccurately monitoring the outbreak.
What is the false positive rate when screening for COVID-19?
This is the million dollar question, and the answer it depends on the prevalence of the virus infection in the population. The lower this is, the more false positive the PCR tests will yield, and the lower the reliability of the test. If the infection rate in the general population is the same number as the test’s false positive rate, the reliability of the positive results received from such a test is essentially zero. Let’s assume that the false positive rate of the current PCR test is in the lower observed range mentioned above, 0.3%. If only 0.3% of the population is truly infected by the virus, the test is pretty much useless to really tell if someone is really infected or not. If 1% of the population is infected and we are screening the population, then still every third person will likely get an inaccurate result. If about 3% of the population is infected with SARS-CoV-2 then still 1 in 10 people would get potentially false information. And so on. This is referred to as test predictive value, and you can see that positive predictive value of the test (test’s ability to accurately predict positive result) is dependant on the prevalence of a disease in a population. And these numbers would be even worse if the false positive rate is higher, which it could be based on past historical data.
The opposite is true for negative results. The higher the prevalence of a disease in a population, the lower the ability of a test to accurately predict the negative result, or the lower the negative predictive value. Positive predictive value of a test has an inverse relation with negative predictive value of a test. While that might be not intuitive at first thought, it actually makes sense. Think of it this way. The more power a test has to identify SARS-CoV-2, the more likely it will identify it when it is not even there. So it will find all the infected people, but it will also find infection in some uninfected people. The more power the test has to confirm a lack of infection by SARS-CoV-2, the less likely it will identify it when it is truly there. So it will accurately identify all the uninfected people, but it will fail to find infection in some infected people. All tests in the world have to strike a best balance between these two performance metrics. Thus the good news is, that if not many people in the population are actually infected, and those who get positive results, it might not be that reliable, those that got a negative result would highly likely get an accurate result. But as soon as the prevalence of SARS-CoV-2 infection increases in the population, we will increase the likelihood that some small portion of those who get negative results could actually be positive for SARS-CoV-2, while increasing the likelihood that positive results are indeed accurate.
Then the real question to ask is, how infected is a given population? This will tell us really how useful the PCR tests are to screen the population for COVID-19 condition.
Are you ready to find out?
Prevalence of SARS-CoV-2 infection
The really best way to establish this type of information is to test for antibodies against SARS-CoV-2 in a population. This is because the presence of antibodies against SARS-CoV-2 in a person means that person was previously infected (or potentially currently infected, depending on what type of antibody you are detecting). Of course, these also will have their own false-positive and false negative rates, but modelling can provide us the best estimates. And you probably will not be surprised to find out that different geographical regions will show different results, as different regions would have seen different rates of infection.
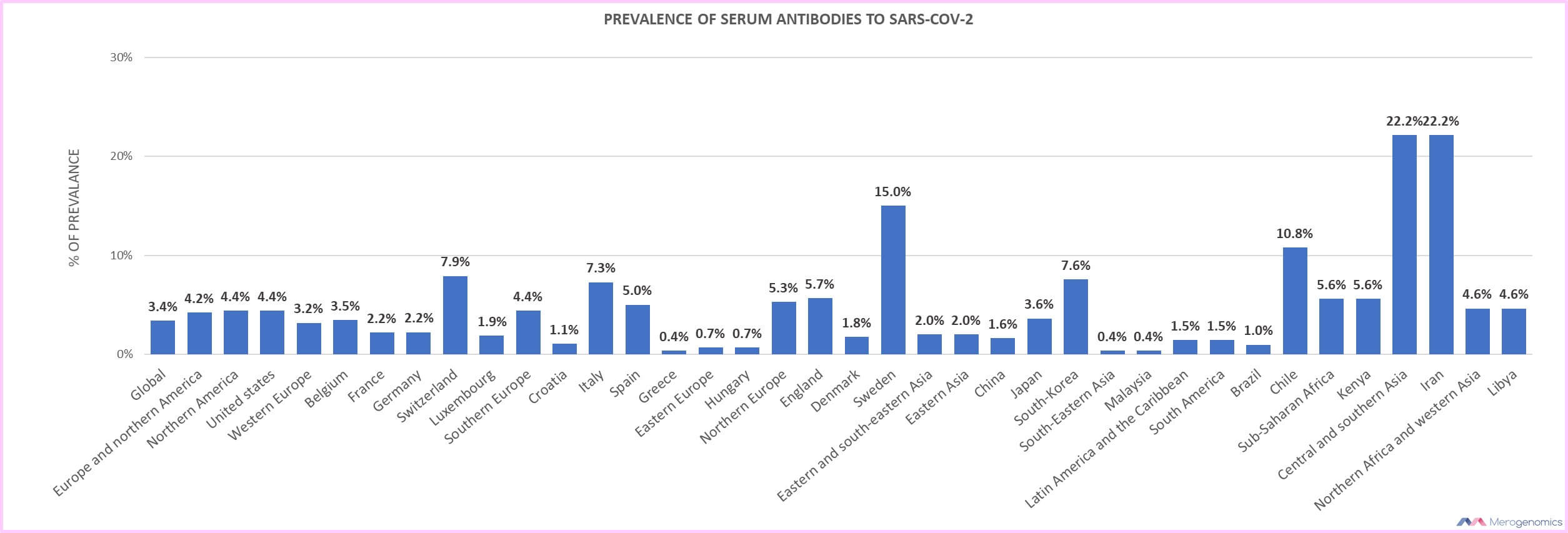
The studies that are best to look at are meta-analysis, a summary of many published studies, and the one we found included nearly 400,000 people from 23 countries (a preprint though, so not yet peer reviewed). The prevalence of SARS-CoV-2 in the general population varied from 0.37% to 22.1%, with a global estimate of 3.38%. That means on that low-end of prevalence the PCR test is potentially yielding uninformative results. Let’s crunch the numbers. If we test 100 people then we would expect positive result 0.37% of the time, but with 0.3% false positive rate of the PCR test, the number would actually be 0.07% and the positive predictive value of the test is only 19% (0.07/0.37)! That means 81% of the time the positive result of the PCR test would be wrong!
Let’s look at the top end of 22.1%. If we test 100 people then we would expect a positive result 22.1% of the time, but with 0.3% false positive rate of the PCR test, that number would actually be 21.8% and the positive predictive value is 98% (21.4/22.1). That means 2% of all results will be wrong. So, you can see how the prevalence of the disease in the population impacts the ability of the test to properly measure outcomes. The reason why is because you are more frequently encountering the condition that the test is designed to identify. If you want to look more into this, we have covered this topic previously in some detail in the discussion of non-invasive prenatal DNA screening that is offered to pregnant women - a topic obviously of great interest to Merogenomics.
Let’s look at one more country, the USA, the country with the most tests and the most reported cases. At the time it had reported 4.1% of SARS-CoV-2 prevalence (more recent study estimates the US SARS-CoV-2 antibody prevalence at 9.3%). If we test 100 people then we would expect positive result 4.1% of the time, but with 0.3% false positive rate of the PCR test, that number would actually be 3.8% and the positive predictive value is 93% (3.8/4.1). That means 7% of all results will be wrong. With more than 12 million confirmed cases in the US right now, we are approaching almost million potentially inaccurate results! But what if the actual false positive rate was 3%? Then 80% of all results will be wrong! Could 3% false positive rate be possible? For sure. We would not know because for some strange reason no one seems to really care to report this crucial statistic, or at least it is not easy to find. How is that even possible? We do not really know.
Below is the breakdown of the SARS-CoV-2 infection prevalence by region. You can do your own calculations for your favourite place in the world that is listed.
Then what are you to do if you are asymptomatic and received positive test results?
Would you be able to get the positive predictive value of the test established by the laboratory? Probably not. Would you be able to obtain the laboratory’s false positive rate? Probably not but that sure would be valuable information, and on top of that it would be valuable how many true negative samples were tested to arrive at this calculation. Would you be able to get Ct value and its explanation? Perhaps and here at least it would give you some ballpark, as low Ct values are much more likely to be indicative of true positive. Without these parameters, any interpretation of the test runs the risk of being unreliable.
Then the first question is, are you at high-risk or low-risk of having the disease prior to that test? Were you exposed to someone with COVID-19? If yes, you should be taking that result very seriously and taking appropriate safety measures.
You could try using one of the approved serological tests. If the results of such a test are also positive then at this point it dramatically increases your odds that indeed you could be an asymptomatic case of COVID-19 and could pose a threat to public safety. You could then consider re-testing with PCR to confirm the diagnosis and quarantine, or quarantine anyway.
If you are in a low-risk category and you tested positive then in theory you are supposed to retest. This is in fact how it has been always done in the past. What we are currently doing during this pandemic is a gross departure from past scientific norms, where a result of a single PCR test which is known to be unreliable in low-risk individuals is deemed enough for a diagnosis. It is difficult to understand why we took this measure, but it might be due to the massive public demand to test as many individuals as possible, and as a consequence we have to cut corners somewhere.
If you do not retest, you could wait to see if symptoms appear. Still, in the least it would be extra prudent to avoid exposure to those who are the most vulnerable, the elderly, in case you are asymptomatic and contagious. If your circumstances allow, you could still impose quarantine for added safety. But should you quarantine without a confirmation of the test? Well, you could have been a false positive! Thus, consider retesting.
And if you got negative result, then indeed you can rest easy. It is very likely an accurate result.
In essence, with the current use of PCR tests for SARS-CoV-2, you can in fact expect that indeed many of them will be false-positives, and the results of these tests should not be driving any policy decisions that govern populations. Not that PCR tests should not be used for screening purposes, but we then have to acknowledge and act accordingly when we deal with screening results versus diagnostic results of the test.
Moving forward, what should we do with COVID-19 testing?
We should definitely continue using the PCR test for diagnostic purposes for those at high risk of COVID-19, as it was intended to be. This is what we started with as the pandemic commenced, and somewhere along the way the world switched to using the test for screening purposes - likely driven by public fear and demand. For those at low risk, we could continue using the PCR test for screening purposes of asymptomatic and presumed healthy populations, but we should redefine the meaning of those results. Those in low Ct values (high viral load) could be treated as a positive cases, but those in high Ct values (low viral load) should be then retested due to the high probability of a false positive. Where that threshold of retesting should be would depend on the prevalence of infection in the population in a given region. Then we would likely be getting a much truer picture of the contagion’s impact. Even better would be for each specific geographical region to conduct viral culturing studies to be better able to define these potential cut-off limits. Each geographical region should also establish and report the false positive rates of all the laboratories used for population screening. Each geographical region should also conduct serological studies to attempt to estimate the prevalence of infection. If so, we actually get to the bottom of the supposed accuracy of these tests and get better grip on the actual meaning of the reported results.
And perhaps we would quiet down at least certain voices in the zealot camps dedicated to fearmongering or dismissing the impact of COVID-19.
This article has been produced by Merogenomics Inc. and edited by Jason Chouinard, B.Sc. Reproduction and reuse of any portion of this content requires Merogenomics Inc. permission and source acknowledgment. It is your responsibility to obtain additional permissions from the third party owners that might be cited by Merogenomics Inc. Merogenomics Inc. disclaims any responsibility for any use you make of content owned by third parties without their permission.
Products and Services Promoted by Merogenomics Inc.
Select target group for DNA testing
Healthy screening |
Undiagnosed diseases |
Cancer |
Prenatal |
Or select popular DNA test
 |
 |
 |
 |
Pharmaco-genetic gene panel |
Non-invasive prenatal screening |
Cancer predisposition gene panel |
Full genome |