Antibodies against SARS-CoV-2
Dr.M.Raszek
Let’s get biological, bio-logical
The coronavirus pandemic is continuing to ravage the world in more ways than we originally could have suspected. It is undoubtedly a defining moment of our lives; for some with tragic consequences but for others it is transformative due to the unusual circumstances in which so many people find themselves. The speed of the infection is so rapid that between each post we see a difference of millions in the number of infected people. And we still do not have any solid treatments or prophylactic measures.
In the last post we started discussing the different options that can be considered when aiming to protect ourselves against COVID-19. We discussed drug development. In this post we will continue with biologics (in the form of antibodies) and vaccine. If you liked the molecular story that unfolded in our last post, you will enjoy this one as well as we desire to showcase the complexity and beauty in the delicate balance of things that exist in this microscopic world.
There has been a tremendous flurry of research trying to set up the production of antibodies that can be used as a weapon against the coronavirus. Antibodies are basically like annoying gluesticks that will latch onto anything that happens to fit their preconfigured shape. We naturally build antibodies against infectious agents that we come into contact with. Thus if someone gets infected with SARS-CoV-2 virus then they will produce antibodies against it. This premise is behind one of the types of tests being developed to determine if someone has actually been infected with the virus - by looking for antibodies against the SARS-CoV-2 coronavirus floating in the patient’s blood. But the big problem with these tests is that they are difficult to develop cheaply for mass testing that can also provide a high level of accuracy. But the scientific world is chasing this hope on a massive scale.
But now imagine, it would be pretty handy if you could have access to such antibodies without the need of infection.
So how do we get our hands on such antibodies that we could use for treatment?
Some of these types of antibodies have already been isolated as an outcome of the prior SARS-CoV outbreak and, due to the structural similarities between the two viruses, some of these old warriors can now be used again against SARS-CoV-2. Other such antibodies could be generated by infecting different animal models then isolating the genes that resulted in new antibody production and then placing those genes into a cellular system, such as bacteria, that can be easily controlled for the mass production of antibodies.
Antibodies can, in theory, also be hijacked from former patients recovering from the infection and then used to benefit another person. This has a clever name to it: convalescent sera. Alas, the kinks of such approach have not yet been worked out but nevertheless, tons of work is ongoing in isolating antibodies from SARS-CoV-2 patients.
Antibodies are also super important for another reason. Understanding how they molecularly target the viral structures can inspire the development of drugs that can take advantage of the same molecular features that are observed at the site of interaction between the antibody and the viral protein that is encountered by the antibody. Think of it as a free gift from nature by showing us viral weak spots. Alternatively, that same knowledge can be taken advantage of to understand how novel vaccines could be designed to stimulate the production of such desired antibodies.
In terms of the use of convalescent sera against SARS-CoV-2 coronavirus, the most advanced work is currently being done by Dr. Arturo Casadevall of John Hopkins Hospital with anti-COVID-19 boost! But going back to the more serious attempts of fighting COVID-19, this approach is also being investigated in infected people to determine if it will keep them from being hospitalized with serious complications. In theory, all you need is a blood donation - a very simple and safe procedure - then the antibodies can be isolated. The only issue is, you will need lots and lots of survivors volunteering their circulatory language of life. We are stuck with a question, will there even be enough volunteers available to start protecting the rest of the population?
Since we do not want to rely on bleeding so many people, we are far more excited about the research of Dr. Horacio Bach of the University of British Columbia, with whom Merogenomics has the honor to collaborate with on a multi-membered, multi-disciplinary project related to COVID-19. Dr. Bach’s expertise is the synthetic production of recombinant antibodies, or basically, pre-designed antibodies that are produced to the specifications of our scientific whims by bacteria. Dr. Bach was recently awarded a Canadian Institutes of Health Research fund to develop a series of antibodies that could neutralize the SARS-CoV-2 by blocking its spike protein from being used to interact with ACE2 receptors. These ACE2 receptors found on the surface of our cells are needed by the virus to enter our cells.
What is cool about the recombinant antibody approach is that it is easily scalable so we would not have to worry about limits of production. In addition, these antibodies can be specialized in their design because we do not have to follow human biological rules. Another important advantage is that antibodies can be generated that are known to already be highly effective. This is in contrast to convalescent sera, where we would be collecting whatever was randomly generated in each individual, and as you will soon see, these can vary in effectiveness.
To listen to Dr. Bach speak about the current coronavirus pandemic check the following interview recorded with him.
In essence, the interest in generating antibodies to fight this disease is quite high as it might turn out to be our first effective means of defense against SARS-CoV-2.
Catch the SARS-CoV-2 with its pants down
Of all the antibodies we have seen reported so far, our favourite has got to be the one labelled CR3022 (antibodies are, after all, just tiny molecular robots. They are huge fans of Star Wars and hence decide to name themselves like their favourite robots movie stars. Not a fact!). Yes, that is so dorky - to have a favourite antibody. But let’s get into a fun biology lesson about antibodies to explain why. And afterwards, let’s see if we have converted you to be a fan of this antibody too.
Discovered in a patient that was infected with the original SARS-CoV, what makes CR3022 so special is how it interacts with the viral spike protein.
As we mentioned in our previous post, the coronavirus spike protein is a homotrimer. This means the final functional product is made up of three identical units put together. Also recall that the spike protein is cleaved by specialized molecular scissors (called furin protease) into two subunits, S1 and S2. But let us add a twist to the plot. The spike protein can exist in multiple conformations. What that means is the protein can shift from one type of shape to another and multiple such options are available for the spike protein. Basically, the SARS-CoV-2 coronavirus spike protein can shape-shift. To be specific, this shape shifting occurs in the S1 subunit of the spike protein - the one that contains the receptor binding domain or RBD, and to be even more specific, it happens right at the apex of the S1 subunit in the area that is at the interface of interaction with the ACE2 human receptor.
These shifts in shape can be divided into a couple categories:
- The “down” conformation: the RBDs is tightly packed against the top of the S2 subunit. This conformation actually prevents binding of the spike protein to ACE2 receptor, as the areas of the RBD needed for this interaction are hidden away. Think of it like a flower that closes its petals at night
- The “up” conformation: the RBDs is jutting out, away from the rest of spike protein, allowing easy interaction with the ACE2 receptor. Think of it as a flower opening its petals in the day
Because the Spike protein is a homotrimer, every single one of the three “amigos” could be either in the “up” or “down” conformation hence you have the possibility of a spike protein experiencing many different conformational options. In the image below, you can see one example where two of the units of a spike protein are in “down” conformation, and one is in “up”.
The color coding is to differentiate the apex of the RBD that is used as the interface to bind the ACE2 receptor in these different conformations.
You may ask yourself, why bother, Mother Nature? Why not just be in the “up” conformation and go for that ACE2 receptor all the way, unicorns and rainbows style (you would have to see our last post to know why unicorns and rainbows)? Well, it is believed that a specific chain of events has to occur for this to play out.
The cleavage of S1 portion of the coronavirus spike protein away from S2 is necessary for the virus membrane to fuse with the membrane of the cell that it is invading. After the cleavage, these subunits remain interacting with one another. This is when the S1 subunit adopts the “up” conformation, binding to the ACE2 receptor. But switching to the “up” conformation and interacting with a receptor has yet another impact. It starts influencing the shape of the S2 subunit which aids in the membrane fusion. When the two subunits are cleaved and this happens, this transition of the S2 from pre-fusion to pro-fusion state is needed for the virus to successfully fuse with the host membrane (our cells). If you pull that trick prior to the cleavage, you will destabilize the complex, initiating a highly embarrassing premature transition toward the pro-fusion state of the spike protein - when the complex is not ready for it, leading to an awkward viral entry failure.
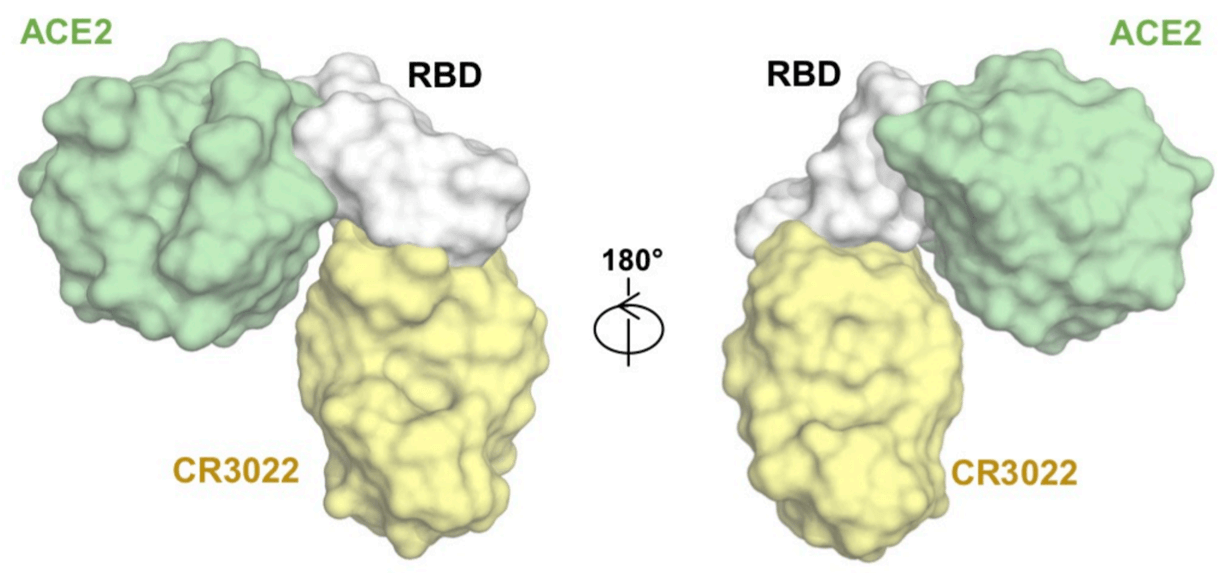
This is finally where our favourite antibody CR3022 enters the picture. Because it is all about where it binds so that it can trap the spike protein in the “up” conformation. CR3022 binds super close to where the human ACE2 receptor also binds, but it does not block the spike protein from binding to the ACE2 receptor. Below you see the two proteins binding to just a fragment of the RBD, with the antibody in yellow and the ACE2 receptor in mint color.
You can see they come pretty close to one another but if you take them away from the picture and take a closer look at where the antibody binds to the spike protein - that is where things get interesting. Below is the picture of spike shown in the “up” conformation on the left-hand side, and “down” conformation on the right-hand side. The area on any molecule that an antibody targets is referred to as an epitope. You can see that in the “up” state, the epitope for the antibody is exposed and the CR3022 antibody can swoop in there and give the spike protein an electromagnetic “kiss of death”. But in the “down” conformation, that epitope is buried, and the CR3022 antibody is left to blindly search for its desired antigen in the vast extracellular environment.
But what we want you to truly understand is how tiny that epitope area actually is. It is just a sliver of a surface area for which the antibody is exquisitely designed to fit perfectly. And unlike the images of drugs you saw in our previous post, where antibodies often occupied deep molecular caverns in the complex shape of these proteins, there is nothing extraordinary about this area that is targeted by CR3022 in terms of its adopted shape. That means this antibody has to be very precisely designed by someone’s body to execute the binding.
And this is just one example of a single antibody. By luck, this one is pretty good because it provides a killer punch to the virus. A type of design that probably would even impress Dr. Bach. But the spike protein does not just sit about in one conformation because with molecules, there is often a bit of fluidity. It can interact with ACE2 for a brief moment, then let go because it has not been cleaved, and so on. So, the moment that a spike protein shows off its horns for an ACE2 receptor, this CR3022 antibody can capture it and not let go, thus the protein plus the virus along with it, is toast.
Au naturelle is not necessarily all belle
The take away from this story should be that natural antibody generation is actually a wildly random process and this person who made this CR3022 antibody when infected with SARS-CoV, well such an outcome might not be seen again. That is because the antibodies are generated through a process referred to as somatic hypermutation. Sounds fancy, but basically somatic means that the DNA mutations are happening in your body sometime after your birth, and therefore it is new information in some cells, now just tiny bit different from the genome you were actually born with (which should be duplicated in all of the daughter cells after the egg is fertilized by a sperm). Cancer mutations are the same type of mutation, somatic mutations, because they randomly occur in the DNA of your cells, and eventually such mutations can accumulate to trigger a negative outcome.
Hypermutation is exactly what it sounds like: DNA mutations occurring at hyper speed (in comparison to what would happen randomly).
We have this somatic hypermutation process built-in on purpose within certain cells that build antibodies so that we can have a fighting chance against the different pathogens that invade us. When the pathogen invasion occurs, some of the cells will attack the pathogen, break it down, and take its components and present it to the cells that make antibodies. Truly, it is just an unabashed exhibition of potential epitopes. Those cells get into action and try to make antibodies that will match any of the presented epitopes. The moment they do match, the winning cells are ordered to make tons more copies of itself to produce an army of antibodies.
But you can see that along the way, it is totally random which epitopes will be exposed, and which antibodies will first be successfully synthesized. As a consequence, you can make an antibody against almost any pathogen surface area that is accessible, and as a further consequence, everyone’s immune reaction can have an entire range of success in terms of clearing the pathogen. Some antibodies will simply be more successful than others.
This brings us to vaccines.
Remember we mentioned in last post that a vaccine is basically a fake infectious agent to trick our bodies into producing an immune reaction that will result in the production and storage of antibodies in our body for when the real infection arrives. This means your body will be requested to fight off this fake infectious agent, and so in the process of building your new immunity you can experience some symptoms similar to actual infection (and yes, even overreaction is possible which could result in death). But usually it is a far milder experience and in return you come equipped with antibodies for whatever nasty bug tries to invade you.
This also means that depending on how a vaccine is designed, and depending on how much available surface area of different antigens are presented as epitopes for antibody production, the vaccination process can lead to very different personalized responses in terms of antibody production. Some of it can be fantastic and some of it can be lackluster. With so much randomness involved in our biology, you should never expect a vaccination program to be 100% effective.
Thus vaccine development is not a trivial process; to be able to design a fake infectious agent that is as safe as possible, tons of trial and error has to take place in the design process, with testing for effectiveness or toxicity first executed on unsuspecting animals. Many animals sacrifice their dear lives so that the rest of us can benefit from medical advances. How slow this complicated process is, will truly be exhibited by the speed of a vaccine development against COVID-19, which you can expect to be one of the fastest ever now that enormous global resources are pooled into fighting this epidemic (although a tiny fraction of what the banks will be expected to contribute in an attempt to “save the economy”. Since we brought up banks in our last post too).
This is why we are particularly excited about the work being done with antibodies and so we think this might the first line of defence we see rolled out to fight the SARS-CoV-2 infection (while more and more people are building their herd immunity as we are already observing).
And for those who will not want to take a vaccine against SARS-CoV-2 (and there will be many), they may have another option available to them besides risking the SARS-CoV-2 infection (and remember, we still do not know the long term consequences of contracting this virus as we are only a scarce few months into the pandemic) - that is to take prophylactic shots of antibodies. Provided they are willing to pay for a new shot every few months.
Or you could just eat apples, drink wine, enjoy chocolate and be merry!
Although we do not expect there will be any studies dedicated to testing these anti-COVID-19 options.
But options are coming, and here you had a glimpse at the molecular world behind the curtains of scientific research that is going on around the world to find a solution to against this coronavirus pandemic.
This article has been produced by Merogenomics Inc. and edited by Jason Chouinard, B.Sc. Reproduction and reuse of any portion of this content requires Merogenomics Inc. permission and source acknowledgment. It is your responsibility to obtain additional permissions from the third party owners that might be cited by Merogenomics Inc. Merogenomics Inc. disclaims any responsibility for any use you make of content owned by third parties without their permission.
Products and Services Promoted by Merogenomics Inc.
Select target group for DNA testing
Healthy screening |
Undiagnosed diseases |
Cancer |
Prenatal |
Or select popular DNA test
 |
 |
 |
 |
Pharmaco-genetic gene panel |
Non-invasive prenatal screening |
Cancer predisposition gene panel |
Full genome |